The d-and f-Block Elements
CBSE · Class 12 · Chemistry
Flashcards for The d-and f-Block Elements — CBSE Class 12 Chemistry. Quick Q&A cards covering key concepts, definitions, and formulas.
Interactive on Super Tutor
Studying The d-and f-Block Elements? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for flashcards and more.
1,000+ Class 12 students started this chapter today
Super Tutor has 15+ illustrations like this for The d-and f-Block Elements alone — flashcards, concept maps, and step-by-step visuals.
See them allWrite the electronic configuration of Chromium (Cr, Z=24) and explain why it deviates from expected configuration.
Answer
Cr: [Ar] 3d⁵ 4s¹ (not 3d⁴ 4s²). Deviation occurs because half-filled d orbitals (d⁵) provide extra stability. The energy difference between 3d and 4s orbitals is small, allowing electron promotion for…
Balance the equation for acidified KMnO₄ oxidizing FeSO₄ and identify the products.
Answer
MnO₄⁻ + 8H⁺ + 5Fe²⁺ → Mn²⁺ + 4H₂O + 5Fe³⁺ Complete: KMnO₄ + 8H₂SO₄ + 5FeSO₄ → K₂SO₄ + MnSO₄ + 5Fe₂(SO₄)₃ + 8H₂O Purple KMnO₄ becomes colorless, green Fe²⁺ becomes yellow Fe³⁺.
Calculate the magnetic moment of Mn²⁺ ion using spin-only formula.
Answer
Mn²⁺: [Ar] 3d⁵ (5 unpaired electrons) Spin-only formula: μ = √[n(n+2)] BM μ = √[5(5+2)] = √35 = 5.92 BM Where n = number of unpaired electrons, BM = Bohr magneton…
Why do transition elements show variable oxidation states? Give examples.
Answer
Due to small energy difference between (n-1)d and ns orbitals, variable numbers of electrons can be lost. Examples: - Fe: +2 (Fe²⁺), +3 (Fe³⁺) - Mn: +2 to +7 (MnO₄⁻) - Cr: +2, +3, +6 (CrO₄²⁻) Maximum …
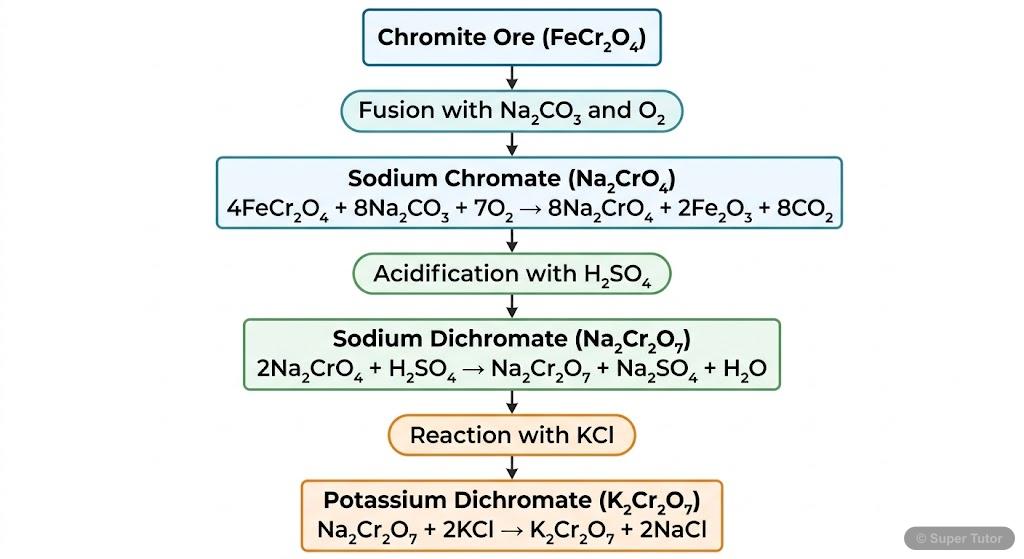
Describe the preparation of K₂Cr₂O₇ from chromite ore (FeCr₂O₄).
Answer
Step 1: 4FeCr₂O₄ + 8Na₂CO₃ + 7O₂ → 8Na₂CrO₄ + 2Fe₂O₃ + 8CO₂ Step 2: 2Na₂CrO₄ + 2H⁺ → Na₂Cr₂O₇ + 2Na⁺ + H₂O Step 3: Na₂Cr₂O₇ + 2KCl → K₂Cr₂O₇ + 2NaCl Orange crystals of K₂Cr₂O₇ crystallize out.
Calculate the equivalent weight of KMnO₄ in acidic medium (Atomic mass: K=39, Mn=55, O=16).
Answer
Molecular weight of KMnO₄ = 39 + 55 + 4(16) = 158 g/mol In acidic medium: MnO₄⁻ + 8H⁺ + 5e⁻ → Mn²⁺ + 4H₂O Electrons gained = 5 Equivalent weight = Molecular weight/5 = 158/5 = 31.6 g/equiv…
What is lanthanoid contraction? How does it affect the properties of 5d transition elements?
Answer
Lanthanoid contraction: Progressive decrease in atomic and ionic radii from La to Lu due to poor shielding by 4f electrons. Effects on 5d elements: - Similar radii to 4d elements (Zr ≈ Hf) - Higher de…
Write the reaction when acidified K₂Cr₂O₇ oxidizes H₂S gas.
Answer
Cr₂O₇²⁻ + 14H⁺ + 6e⁻ → 2Cr³⁺ + 7H₂O (reduction half) 3H₂S → 6H⁺ + 3S + 6e⁻ (oxidation half) Overall: K₂Cr₂O₇ + 4H₂SO₄ + 3H₂S → K₂SO₄ + Cr₂(SO₄)₃ + 7H₂O + 3S↓ Sulfur precipitates as yellow solid.
+12 more flashcards available
Practice AllFrequently Asked Questions
What are the important topics in The d-and f-Block Elements for CBSE Class 12 Chemistry?
How to score full marks in The d-and f-Block Elements — CBSE Class 12 Chemistry?
How many flashcards are available for The d-and f-Block Elements?
Sources & Official References
- NCERT Official — ncert.nic.in
- CBSE Academic — cbseacademic.nic.in
- CBSE Official — cbse.gov.in
- National Education Policy 2020 — education.gov.in
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for The d-and f-Block Elements
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full The d-and f-Block Elements chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for CBSE Class 12 Chemistry.