Matter in Our Surroundings
CBSE · Class 9 · Science
Quick revision notes for Matter in Our Surroundings — CBSE Class 9 Science. Key concepts, formulas, and definitions for last-minute revision.
Interactive on Super Tutor
Studying Matter in Our Surroundings? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for revision notes and more.
1,000+ Class 9 students started this chapter today
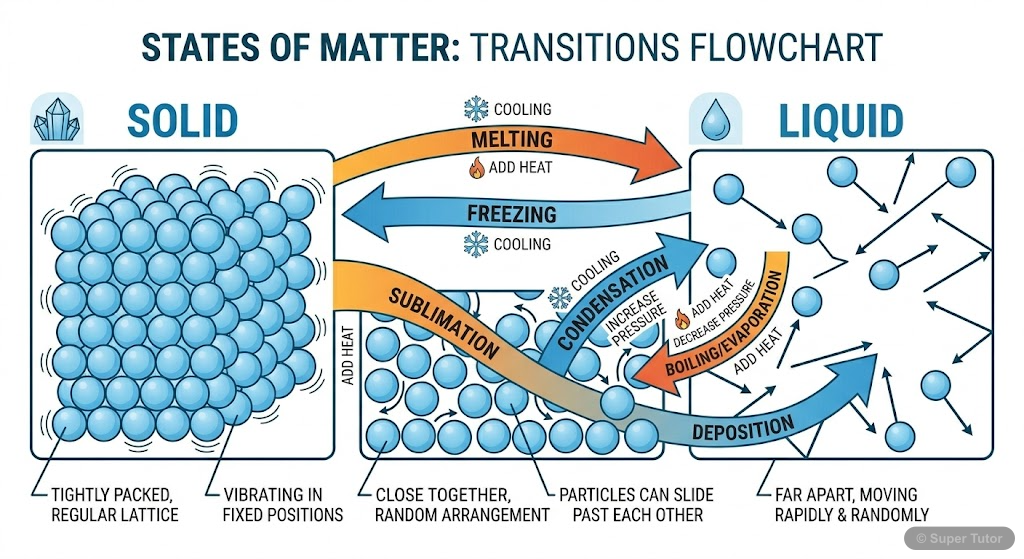
This is just one of 16+ visuals inside Super Tutor's Matter in Our Surroundings chapter
Explore the full setKey Topics to Revise
Physical Nature of Matter
- Matter is made up of tiny particles - this was proven through dissolution experiments with salt/sugar in water
- Particles of matter are extremely small - demonstrated by potassium permanganate dilution experiment where few crystals can color 1000L of water
- Ancient Indian philosophers classified matter as 'Panch Tatva' - air, earth, fire, sky, and water
Characteristics of Particles of Matter
- Particles have space between them - shown by sugar/salt dissolving in water without increasing water level
- Particles are in continuous motion (kinetic energy) - increases with temperature
- Particles attract each other with varying force strength - strongest in solids, weakest in gases
States of Matter
- Three states: Solid, Liquid, Gas - differ in particle arrangement and movement
- SOLIDS: Fixed shape, fixed volume, negligible compressibility, particles closely packed with strong attraction
- LIQUIDS: No fixed shape, fixed volume, particles can move freely but still attracted to each other
Change of State
- States can change by altering temperature or pressure
- MELTING: Solid to liquid at melting point (273 K for ice)
- BOILING: Liquid to gas at boiling point (373 K for water)
Get complete notes with diagrams and examples
Full NotesKey Concepts
Frequently Asked Questions
What are the important topics in Matter in Our Surroundings for CBSE Class 9 Science?
How to score full marks in Matter in Our Surroundings — CBSE Class 9 Science?
Sources & Official References
- NCERT Official — ncert.nic.in
- CBSE Academic — cbseacademic.nic.in
- CBSE Official — cbse.gov.in
- National Education Policy 2020 — education.gov.in
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Matter in Our Surroundings
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Study Plan
Step-by-step plan to ace this chapter
Flashcards
Quick-fire cards for active recall
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Matter in Our Surroundings chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for CBSE Class 9 Science.