Some Basic Concepts Of Chemistry — Practice Questions
AP EAPCET · Chemistry
Practice questions from Some Basic Concepts Of Chemistry for AP EAPCET Chemistry. Test your understanding with MCQs and problem sets.
Interactive on Super Tutor
Studying Some Basic Concepts Of Chemistry? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for AP EAPCET Chemistry.
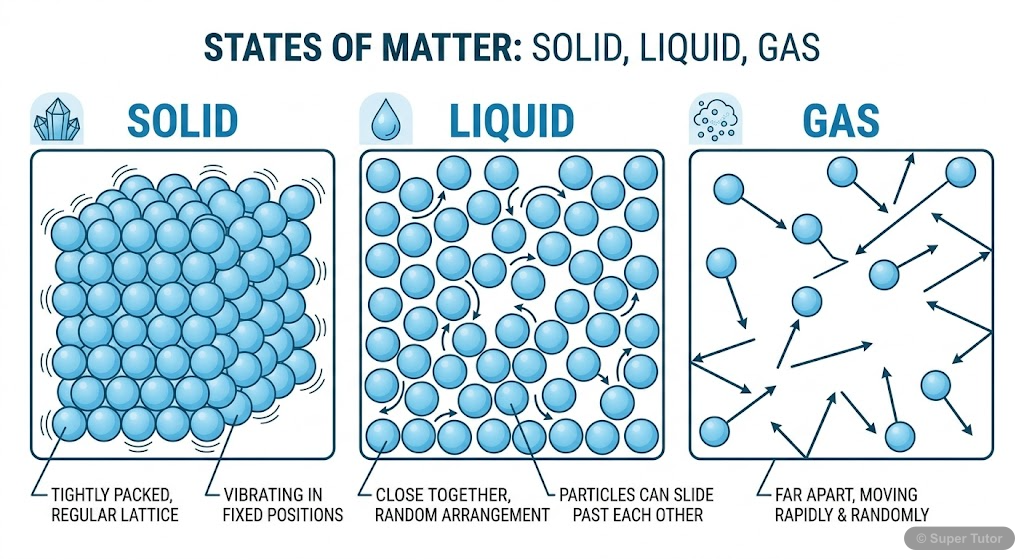
Super Tutor has 16+ illustrations like this for Some Basic Concepts Of Chemistry alone — flashcards, concept maps, and step-by-step visuals.
See them allPractice Questions — Some Basic Concepts Of Chemistry
340 questions available from Some Basic Concepts Of Chemistry for AP EAPCET Chemistry.
Question Types
Sample Questions
What is the mass of one mole of carbon dioxide (CO₂)? (Given: C = 12 u, O = 16 u)
According to Avogadro's law, equal volumes of gases at the same temperature and pressure contain:
Which of the following represents the empirical formula of glucose (C₆H₁₂O₆)?
What is the molarity of a solution containing 4 g of NaOH dissolved in 500 mL of solution? (Given: Na = 23, O = 16, H = 1)
+336 more questions — practice in Super Tutor
Frequently Asked Questions
What topics are covered in Some Basic Concepts Of Chemistry for AP EAPCET?
How important is Some Basic Concepts Of Chemistry for AP EAPCET?
How to prepare Some Basic Concepts Of Chemistry for AP EAPCET?
More resources for Some Basic Concepts Of Chemistry
For AP EAPCET aspirants
Get the full Some Basic Concepts Of Chemistry chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for AP EAPCET Chemistry.