Chapter 17 of 19
Syllabus
Hydrocarbons — Syllabus
AP EAPCET · Chemistry
Topics covered in Hydrocarbons for AP EAPCET Chemistry. Understand the syllabus structure and key areas to focus on.
Interactive on Super Tutor
Studying Hydrocarbons? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for AP EAPCET Chemistry.
Super Tutor
This is just one of 28+ visuals inside Super Tutor's Hydrocarbons chapter
Explore the full setHydrocarbons — Syllabus & Topics
Topics covered in Hydrocarbons for AP EAPCET Chemistry.
Topics in Hydrocarbons
1
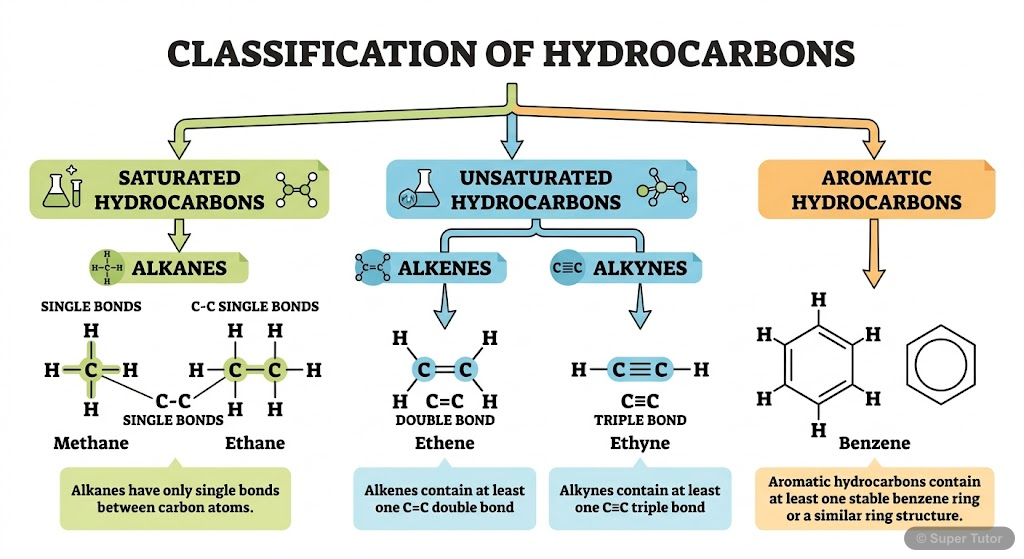
9.1 Classification of Hydrocarbons
- Hydrocarbons are classified into three main categories: Saturated (alkanes and cycloalkanes), Unsaturated (alkenes and alkynes), and Aromatic (benzene derivatives)
- Saturated hydrocarbons contain only single C-C bonds and follow the formula CnH2n+2 for alkanes and CnH2n for cycloalkanes
- Unsaturated hydrocarbons contain double or triple bonds: alkenes (C=C) have formula CnH2n, alkynes (C≡C) have formula CnH2n-2
2
9.2 Alkanes - Structure and Properties
- Alkanes have sp³ hybridized carbon atoms with tetrahedral geometry (bond angle 109°28')
- All C-C bonds are 1.54 Å long and C-H bonds are 1.09 Å long
- Alkanes exhibit chain isomerism starting from C₄H₁₀ (butane has 2 isomers, pentane has 3, hexane has 5)
3
9.2 Alkane Preparation Methods
- Hydrogenation of alkenes/alkynes: R-CH=CH-R' + H₂ → R-CH₂-CH₂-R' (Pt/Pd/Ni catalyst)
- Reduction of alkyl halides: R-X + H₂ → R-H + HX (Zn/HCl)
- Wurtz reaction: 2R-X + 2Na → R-R + 2NaX (dry ether, gives even-carbon alkanes)
4
9.2 Alkane Chemical Reactions
- Halogenation follows free radical mechanism: Initiation (Cl₂ → 2Cl•), Propagation (CH₄ + Cl• → CH₃• + HCl), Termination (Cl• + Cl• → Cl₂)
- Halogenation reactivity order: F₂ > Cl₂ > Br₂ > I₂ (fluorination too violent, iodination needs oxidizing agent)
- Hydrogen replacement order: 3° > 2° > 1° (tertiary hydrogen most easily replaced)
Key Concepts
Hydrocarbons are classified into three mainAlkanes have general formula CₙH₂ₙ₊₂Alkenes have general formula CₙH₂ₙMarkovnikov's rule states that inAlkynes with general formula CₙH₂ₙ₋₂ have
Frequently Asked Questions
What topics are covered in Hydrocarbons for AP EAPCET?
Hydrocarbons is an important chapter in AP EAPCET Chemistry. It covers key concepts and formulas that are frequently tested in the exam. Key topics include: 9.1 Classification of Hydrocarbons, 9.2 Alkanes - Structure and Properties, 9.2 Alkane Preparation Methods, 9.2 Alkane Chemical Reactions.
How important is Hydrocarbons for AP EAPCET?
Hydrocarbons is a frequently tested chapter in AP EAPCET Chemistry. Questions from this chapter appear regularly in previous year papers. There are 267 practice questions available for this chapter.
How to prepare Hydrocarbons for AP EAPCET?
Start by understanding the core concepts, then solve practice questions. Focus on formulas and their applications. Use revision notes for quick review before the exam.
More resources for Hydrocarbons
For AP EAPCET aspirants
Get the full Hydrocarbons chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for AP EAPCET Chemistry.