Chapter 9 of 19
Syllabus
Thermodynamics — Syllabus
BCECE · Chemistry
Topics covered in Thermodynamics for BCECE Chemistry. Understand the syllabus structure and key areas to focus on.
Interactive on Super Tutor
Studying Thermodynamics? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for BCECE Chemistry.
Super Tutor
Super Tutor has 5+ illustrations like this for Thermodynamics alone — flashcards, concept maps, and step-by-step visuals.
See them allThermodynamics — Syllabus & Topics
Topics covered in Thermodynamics for BCECE Chemistry.
Topics in Thermodynamics
1
5.1 Thermodynamic Terms and Systems
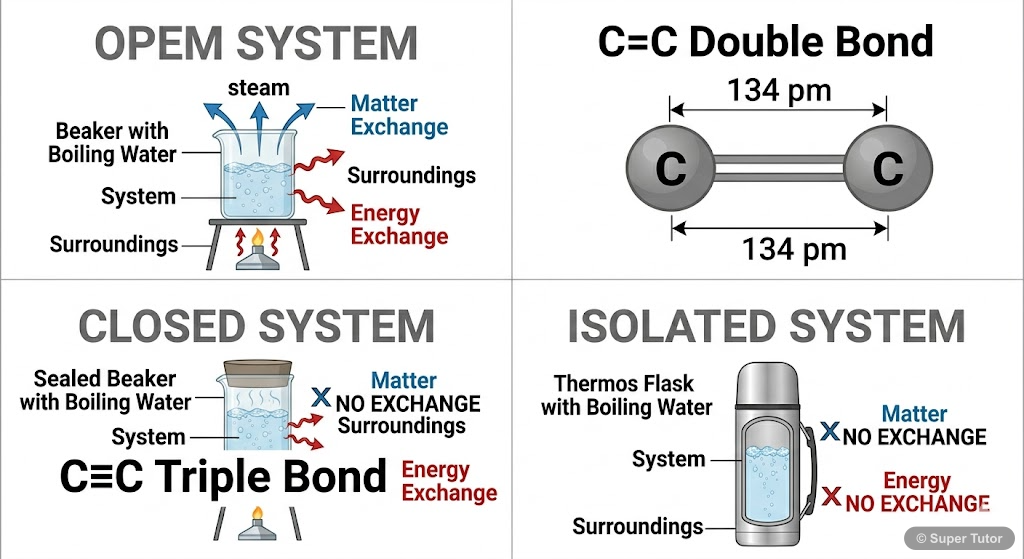
- A thermodynamic system is the part of the universe under study, while surroundings constitute the rest of the universe
- Open system: Exchange of both matter and energy with surroundings (e.g., boiling water in open pan)
- Closed system: Exchange of energy only, no matter exchange (e.g., water in sealed bottle)
2
5.2 Applications - Work and Heat Calculations
- For isothermal reversible expansion: w = -nRT ln(Vf/Vi) = -2.303nRT log(Vf/Vi)
- For free expansion (expansion into vacuum): w = 0
- For adiabatic process: q = 0, so ΔU = w
3
5.3 Enthalpy and Calorimetry
- Enthalpy H = U + PV (extensive property and state function)
- At constant pressure: qp = ΔH
- At constant volume: qv = ΔU
4
5.4 Standard Enthalpy Changes and Hess's Law
- Standard conditions: 298K, 1 bar pressure, pure substances
- Standard enthalpy of formation (Δf H°): enthalpy change when 1 mol compound formed from elements
- Standard enthalpy of formation of elements in standard state = 0
Key Concepts
A thermodynamic system is the partEnergy cannot be created or destroyedEnthalpy is the total heat contentWork done by gas during expansionDifferent enthalpy types
Frequently Asked Questions
What topics are covered in Thermodynamics for BCECE?
Thermodynamics is an important chapter in BCECE Chemistry. It covers key concepts and formulas that are frequently tested in the exam. Key topics include: 5.1 Thermodynamic Terms and Systems, 5.2 Applications - Work and Heat Calculations, 5.3 Enthalpy and Calorimetry, 5.4 Standard Enthalpy Changes and Hess's Law.
How important is Thermodynamics for BCECE?
Thermodynamics is a frequently tested chapter in BCECE Chemistry. Questions from this chapter appear regularly in previous year papers. There are 269 practice questions available for this chapter.
How to prepare Thermodynamics for BCECE?
Start by understanding the core concepts, then solve practice questions. Focus on formulas and their applications. Use revision notes for quick review before the exam.
More resources for Thermodynamics
For BCECE aspirants
Get the full Thermodynamics chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for BCECE Chemistry.