Chapter 6 of 19
Revision Notes
Chemical Kinetics — Revision Notes
BITSAT · Chemistry
Quick revision notes for Chemical Kinetics — key concepts, formulas, and definitions for BITSAT Chemistry preparation.
Interactive on Super Tutor
Studying Chemical Kinetics? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for BITSAT Chemistry.
Super Tutor
Super Tutor has 16+ illustrations like this for Chemical Kinetics alone — flashcards, concept maps, and step-by-step visuals.
See them allRevision Notes — Chemical Kinetics
Key concepts, formulas, and definitions from Chemical Kinetics for BITSAT Chemistry preparation.
Key Topics to Revise
1
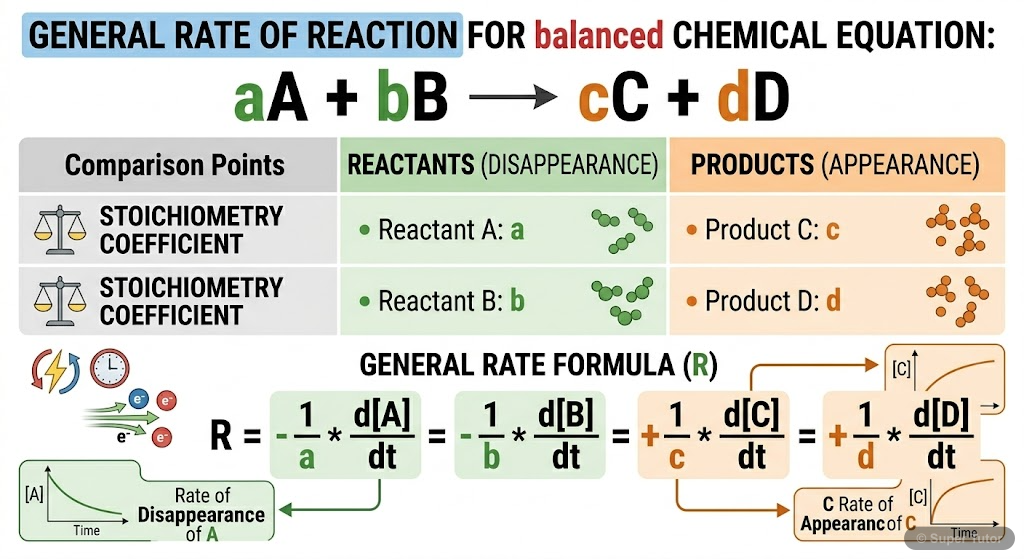
3.1 Rate of Chemical Reaction
- Rate of reaction is defined as the change in concentration of reactant or product per unit time
- For reaction R → P: Rate = -Δ[R]/Δt = +Δ[P]/Δt (negative for reactants, positive for products)
- Units: mol L⁻¹ s⁻¹ or atm s⁻¹ for gaseous reactions
2
3.2 Factors Affecting Reaction Rate & Rate Laws
- Concentration: Higher concentration → faster reaction (more collisions)
- Temperature: ~10°C rise doubles reaction rate for most reactions
- Physical state: Gas > Liquid > Solid (molecular mobility)
3
3.3 Integrated Rate Equations - Zero Order
- Zero order: Rate independent of concentration (Rate = k)
- Examples: NH₃ decomposition on Pt surface at 1130K
- Rate remains constant throughout reaction
4
3.3 Integrated Rate Equations - First Order
- First order: Rate proportional to one concentration term
- Examples: N₂O₅ decomposition, radioactive decay
- Integrated equation: k = (2.303/t) log([R]₀/[R]t)
Get complete revision notes with diagrams and examples — continue in Super Tutor
Key Concepts
The rate of reaction is definedSix main factors influence reaction ratesRate law expresses reaction rateOrder is the sum of exponentsThese relate concentration to time
Frequently Asked Questions
What topics are covered in Chemical Kinetics for BITSAT?
Chemical Kinetics is an important chapter in BITSAT Chemistry. It covers key concepts and formulas that are frequently tested in the exam. Key topics include: 3.1 Rate of Chemical Reaction, 3.2 Factors Affecting Reaction Rate & Rate Laws, 3.3 Integrated Rate Equations - Zero Order, 3.3 Integrated Rate Equations - First Order.
How important is Chemical Kinetics for BITSAT?
Chemical Kinetics is a frequently tested chapter in BITSAT Chemistry. Questions from this chapter appear regularly in previous year papers. There are 276 practice questions available for this chapter.
How to prepare Chemical Kinetics for BITSAT?
Start by understanding the core concepts, then solve practice questions. Focus on formulas and their applications. Use revision notes for quick review before the exam.
More resources for Chemical Kinetics
For BITSAT aspirants
Get the full Chemical Kinetics chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for BITSAT Chemistry.