Chapter 13 of 19
Revision Notes
Redox Reactions — Revision Notes
BITSAT · Chemistry
Quick revision notes for Redox Reactions — key concepts, formulas, and definitions for BITSAT Chemistry preparation.
Interactive on Super Tutor
Studying Redox Reactions? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for BITSAT Chemistry.
Super Tutor
This is just one of 12+ visuals inside Super Tutor's Redox Reactions chapter
Explore the full setRevision Notes — Redox Reactions
Key concepts, formulas, and definitions from Redox Reactions for BITSAT Chemistry preparation.
Key Topics to Revise
1
Classical Concept of Oxidation and Reduction
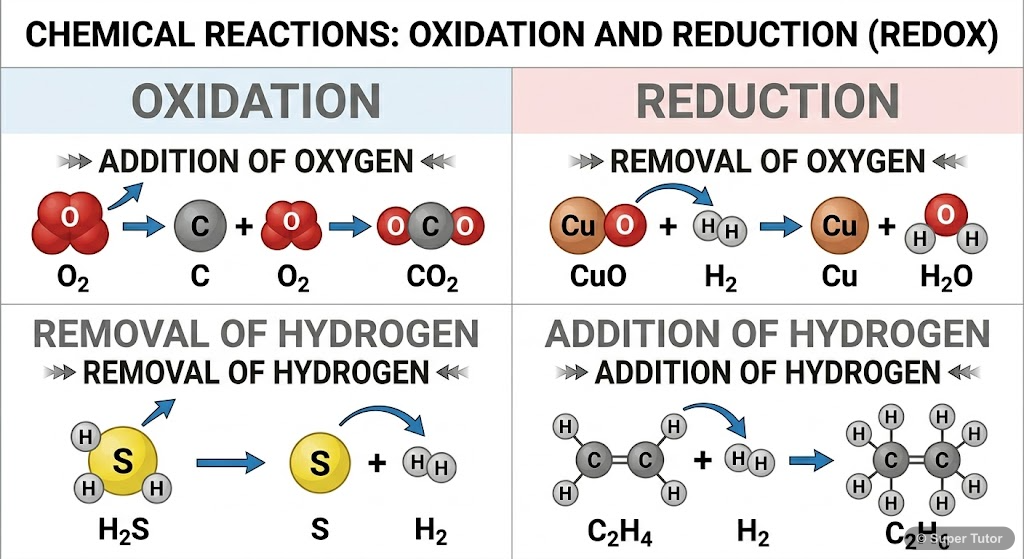
- Oxidation: Addition of oxygen/electronegative element OR removal of hydrogen/electropositive element
- Reduction: Removal of oxygen/electronegative element OR addition of hydrogen/electropositive element
- Example: 2Mg(s) + O₂(g) → 2MgO(s) (Oxidation of Mg)
2
Oxidation Numbers and Rules
- Oxidation number = charge an atom appears to have in combined state
- Free elements have oxidation number = 0
- Monoatomic ions: oxidation number = ionic charge
3
Types of Redox Reactions
- Combination: A + B → AB (at least one element in free state)
- Decomposition: AB → A + B (at least one product in free state)
- Displacement: AB + C → AC + B (more reactive displaces less reactive)
4
Balancing Redox Equations
- Two methods: Oxidation Number Method and Half-Reaction Method
- Oxidation Number Method: Balance atoms with changed oxidation states first
- Half-Reaction Method: Separate into oxidation and reduction half-reactions
Get complete revision notes with diagrams and examples — continue in Super Tutor
Key Concepts
OriginallyModern definitionOxidation number is the hypothetical charge1) CombinationTwo methods
Frequently Asked Questions
What topics are covered in Redox Reactions for BITSAT?
Redox Reactions is an important chapter in BITSAT Chemistry. It covers key concepts and formulas that are frequently tested in the exam. Key topics include: Classical Concept of Oxidation and Reduction, Oxidation Numbers and Rules, Types of Redox Reactions, Balancing Redox Equations.
How important is Redox Reactions for BITSAT?
Redox Reactions is a frequently tested chapter in BITSAT Chemistry. Questions from this chapter appear regularly in previous year papers. There are 172 practice questions available for this chapter.
How to prepare Redox Reactions for BITSAT?
Start by understanding the core concepts, then solve practice questions. Focus on formulas and their applications. Use revision notes for quick review before the exam.
More resources for Redox Reactions
For BITSAT aspirants
Get the full Redox Reactions chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for BITSAT Chemistry.