Chapter 4 of 19
Syllabus
Electrochemistry — Syllabus
BITSAT · Chemistry
Topics covered in Electrochemistry for BITSAT Chemistry. Understand the syllabus structure and key areas to focus on.
Interactive on Super Tutor
Studying Electrochemistry? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for BITSAT Chemistry.
Super Tutor
This is just one of 17+ visuals inside Super Tutor's Electrochemistry chapter
Explore the full setElectrochemistry — Syllabus & Topics
Topics covered in Electrochemistry for BITSAT Chemistry.
Topics in Electrochemistry
1
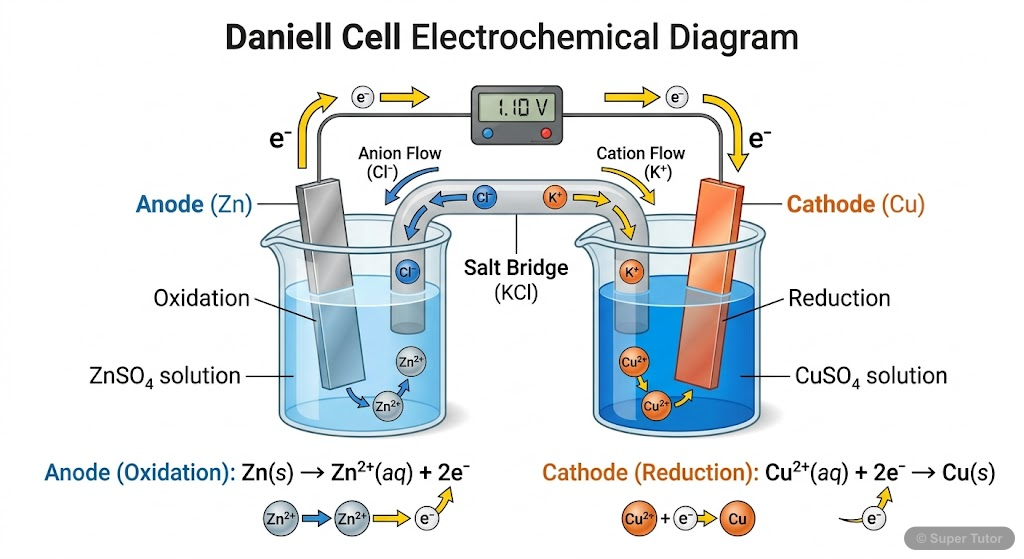
Electrochemical Cells and Galvanic Cells
- Electrochemical cells convert chemical energy into electrical energy through spontaneous redox reactions
- A galvanic cell consists of two half-cells connected by a salt bridge and external circuit
- Anode is the negative electrode where oxidation occurs (electrons are released)
2
Nernst Equation and Concentration Effects
- Nernst equation relates cell potential to concentration of species involved
- At equilibrium, Ecell = 0 and can be used to find equilibrium constant
- Cell potential decreases as the cell reaction approaches equilibrium
3
Conductance of Electrolytic Solutions
- Electrical conductivity depends on the presence and mobility of ions in solution
- Conductance (G) is the reciprocal of resistance (R): G = 1/R
- Conductivity (κ) is the conductance of a solution of unit cross-sectional area and unit length
4
Electrolysis and Faraday's Laws
- Electrolysis uses electrical energy to drive non-spontaneous chemical reactions
- Products depend on electrode material, electrolyte concentration, and applied potential
- Faraday's first law: Amount of substance liberated is proportional to charge passed
Key Concepts
Electrochemical (galvanic) cells convert chemical energyStandard electrode potential (E°) is measuredE = E°Conductance G = 1/R = κA/lΛₘ° = ν₊λ₊° + ν₋λ₋°
Frequently Asked Questions
What topics are covered in Electrochemistry for BITSAT?
Electrochemistry is an important chapter in BITSAT Chemistry. It covers key concepts and formulas that are frequently tested in the exam. Key topics include: Electrochemical Cells and Galvanic Cells, Nernst Equation and Concentration Effects, Conductance of Electrolytic Solutions, Electrolysis and Faraday's Laws.
How important is Electrochemistry for BITSAT?
Electrochemistry is a frequently tested chapter in BITSAT Chemistry. Questions from this chapter appear regularly in previous year papers. There are 271 practice questions available for this chapter.
How to prepare Electrochemistry for BITSAT?
Start by understanding the core concepts, then solve practice questions. Focus on formulas and their applications. Use revision notes for quick review before the exam.
More resources for Electrochemistry
For BITSAT aspirants
Get the full Electrochemistry chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for BITSAT Chemistry.