Some Basic Concepts Of Chemistry — Revision Notes
CUET (UG) · Chemistry
Quick revision notes for Some Basic Concepts Of Chemistry — key concepts, formulas, and definitions for CUET (UG) Chemistry preparation.
Interactive on Super Tutor
Studying Some Basic Concepts Of Chemistry? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for CUET (UG) Chemistry.
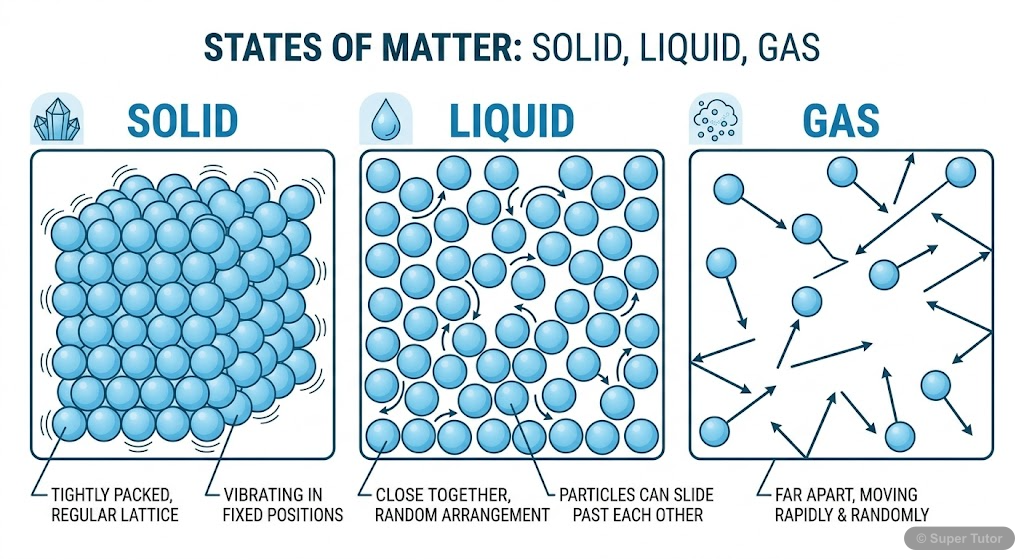
Super Tutor has 16+ illustrations like this for Some Basic Concepts Of Chemistry alone — flashcards, concept maps, and step-by-step visuals.
See them allRevision Notes — Some Basic Concepts Of Chemistry
Key concepts, formulas, and definitions from Some Basic Concepts Of Chemistry for CUET (UG) Chemistry preparation.
Key Topics to Revise
Nature of Matter and Classification
- Matter is anything that occupies space and has mass
- Three physical states: solid (fixed shape and volume), liquid (fixed volume, takes container shape), gas (neither fixed shape nor volume)
- States are interconvertible: Solid ⇌ Liquid ⇌ Gas (heating/cooling)
SI Units and Measurement
- Seven SI base units: length (m), mass (kg), time (s), electric current (A), temperature (K), amount of substance (mol), luminous intensity (cd)
- Common prefixes: nano (10⁻⁹), micro (10⁻⁶), milli (10⁻³), centi (10⁻²), kilo (10³), mega (10⁶)
- Volume units: 1 L = 1000 mL = 1000 cm³ = 1 dm³ = 10⁻³ m³
Significant Figures and Scientific Notation
- Scientific notation: N × 10ⁿ where 1 ≤ N < 10 and n is integer
- Significant figures: All non-zero digits, zeros between non-zero digits, trailing zeros after decimal point
- Leading zeros are NOT significant
Laws of Chemical Combination
- Law of Conservation of Mass: Mass neither created nor destroyed in chemical reactions
- Law of Definite Proportions: Elements in compound always present in fixed mass ratio
- Law of Multiple Proportions: When two elements form multiple compounds, mass ratios are simple whole numbers
Get complete revision notes with diagrams and examples — continue in Super Tutor
Key Concepts
Frequently Asked Questions
What topics are covered in Some Basic Concepts Of Chemistry for CUET (UG)?
How important is Some Basic Concepts Of Chemistry for CUET (UG)?
How to prepare Some Basic Concepts Of Chemistry for CUET (UG)?
More resources for Some Basic Concepts Of Chemistry
Syllabus
Some Basic Concepts Of Chemistry — syllabus
Important Topics
Some Basic Concepts Of Chemistry — important topics
Practice Questions
Some Basic Concepts Of Chemistry — practice questions
Study Plan
Some Basic Concepts Of Chemistry — study plan
Formula Sheet
Some Basic Concepts Of Chemistry — formula sheet
For CUET (UG) aspirants
Get the full Some Basic Concepts Of Chemistry chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for CUET (UG) Chemistry.