Chemical Equilibrium — Practice Questions
JEE Advanced · Chemistry
Practice questions from Chemical Equilibrium for JEE Advanced Chemistry. Test your understanding with MCQs and problem sets.
Interactive on Super Tutor
Studying Chemical Equilibrium? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for JEE Advanced Chemistry.
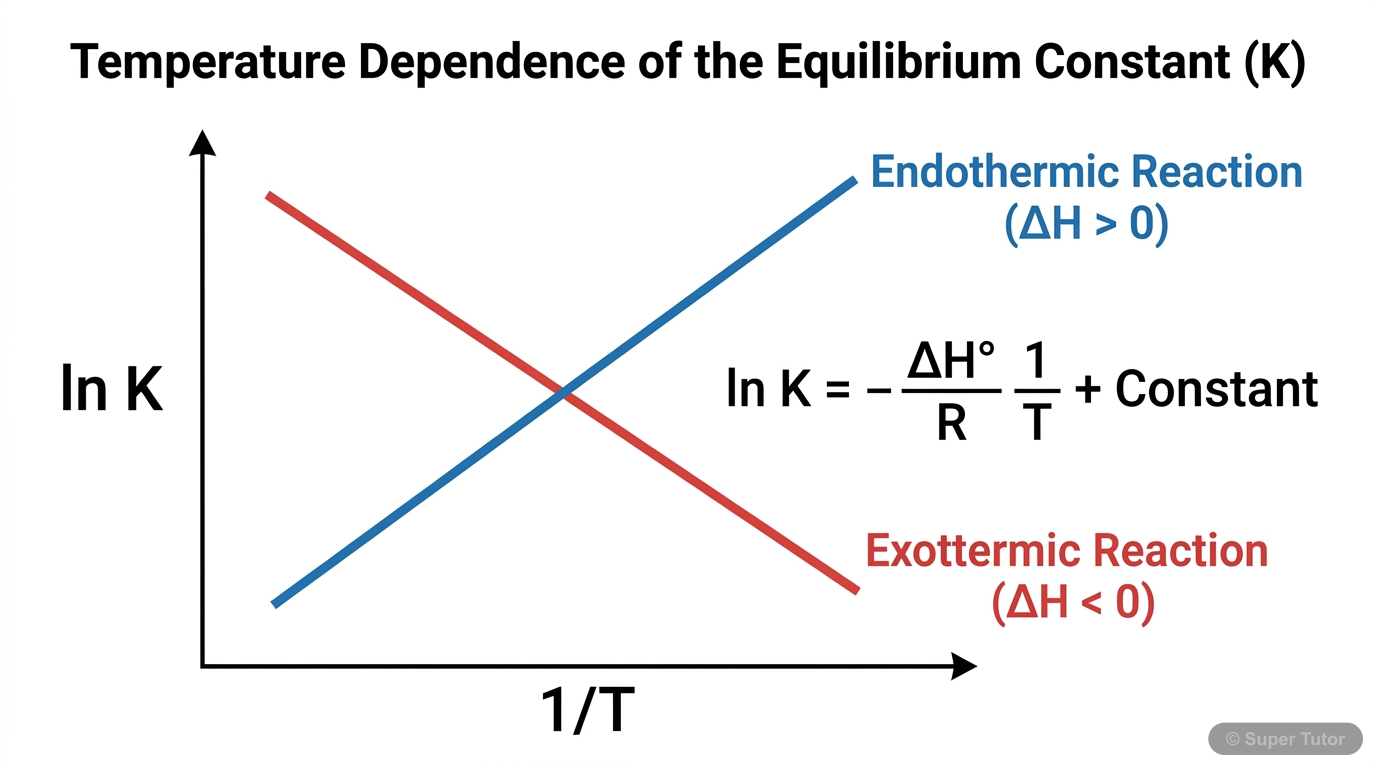
This is just one of 11+ visuals inside Super Tutor's Chemical Equilibrium chapter
Explore the full setPractice Questions — Chemical Equilibrium
72 questions available from Chemical Equilibrium for JEE Advanced Chemistry.
Question Types
Sample Questions
What is the equilibrium constant expression (Kc) for the reaction: 2SO₂(g) + O₂(g) ⇌ 2SO₃(g)?
According to Le Chatelier's principle, what happens when temperature is increased for the exothermic reaction: N₂(g) + 3H₂(g) ⇌ 2NH₃(g) + heat?
For a reaction at equilibrium, if Kc = 64, what can we conclude about the reaction?
What is the relationship between Kp and Kc for the reaction: H₂(g) + I₂(g) ⇌ 2HI(g)?
+68 more questions — practice in Super Tutor
Frequently Asked Questions
What topics are covered in Chemical Equilibrium for JEE Advanced?
How important is Chemical Equilibrium for JEE Advanced?
How to prepare Chemical Equilibrium for JEE Advanced?
More resources for Chemical Equilibrium
For JEE Advanced aspirants
Get the full Chemical Equilibrium chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for JEE Advanced Chemistry.