Chapter 40 of 46
Revision Notes
Alcohols, Phenols and Ethers — Revision Notes
JEE Advanced · Chemistry
Quick revision notes for Alcohols, Phenols and Ethers — key concepts, formulas, and definitions for JEE Advanced Chemistry preparation.
Interactive on Super Tutor
Studying Alcohols, Phenols and Ethers? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for JEE Advanced Chemistry.
Super Tutor
This is just one of 14+ visuals inside Super Tutor's Alcohols, Phenols and Ethers chapter
Explore the full setRevision Notes — Alcohols, Phenols and Ethers
Key concepts, formulas, and definitions from Alcohols, Phenols and Ethers for JEE Advanced Chemistry preparation.
Key Topics to Revise
1
Classification and Nomenclature
- Alcohols are classified as primary (1°), secondary (2°), or tertiary (3°) based on the carbon atom bearing the -OH group
- Phenols have -OH group directly attached to benzene ring, making them more acidic than alcohols
- Ethers can be symmetrical (R-O-R) or unsymmetrical (R-O-R') and are named using alkoxy nomenclature
2
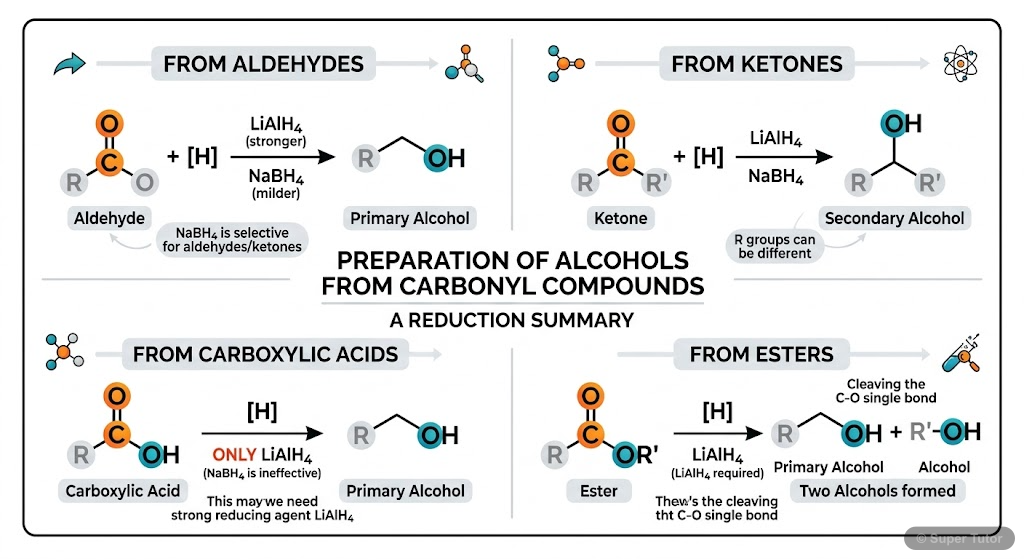
Preparation Methods
- Alcohols from alkenes: Hydration (Markovnikov), Oxymercuration-demercuration, Hydroboration-oxidation (anti-Markovnikov)
- Alcohols from carbonyl compounds: Reduction of aldehydes/ketones, Grignard reactions
- Phenols from diazonium salts, benzene sulfonates, cumene oxidation
3
Physical Properties and Hydrogen Bonding
- Hydrogen bonding increases boiling points: alcohols > ethers > alkanes of similar MW
- Solubility in water decreases with increasing chain length due to hydrophobic alkyl groups
- Phenols have higher boiling points than alcohols due to aromatic stabilization and H-bonding
4
Chemical Reactions of Alcohols
- Reaction with metals (Na, K) to form alkoxides and H₂ gas
- Oxidation: 1° alcohols → aldehydes → carboxylic acids; 2° alcohols → ketones; 3° alcohols resist oxidation
- Dehydration with conc. H₂SO₄: 3° > 2° > 1° alcohols, follows Saytzeff's rule
Get complete revision notes with diagrams and examples — continue in Super Tutor
Key Concepts
Alcohols are classified based onKey preparation methods includeAcidic strength orderMajor reactions includeR
Frequently Asked Questions
What topics are covered in Alcohols, Phenols and Ethers for JEE Advanced?
Alcohols, Phenols and Ethers is an important chapter in JEE Advanced Chemistry. It covers key concepts and formulas that are frequently tested in the exam. Key topics include: Classification and Nomenclature, Preparation Methods, Physical Properties and Hydrogen Bonding, Chemical Reactions of Alcohols.
How important is Alcohols, Phenols and Ethers for JEE Advanced?
Alcohols, Phenols and Ethers is a frequently tested chapter in JEE Advanced Chemistry. Questions from this chapter appear regularly in previous year papers. There are 75 practice questions available for this chapter.
How to prepare Alcohols, Phenols and Ethers for JEE Advanced?
Start by understanding the core concepts, then solve practice questions. Focus on formulas and their applications. Use revision notes for quick review before the exam.
More resources for Alcohols, Phenols and Ethers
For JEE Advanced aspirants
Get the full Alcohols, Phenols and Ethers chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for JEE Advanced Chemistry.