Chemical Bonding and Molecular Structure — Revision Notes
JEE Advanced · Chemistry
Quick revision notes for Chemical Bonding and Molecular Structure — key concepts, formulas, and definitions for JEE Advanced Chemistry preparation.
Interactive on Super Tutor
Studying Chemical Bonding and Molecular Structure? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for JEE Advanced Chemistry.
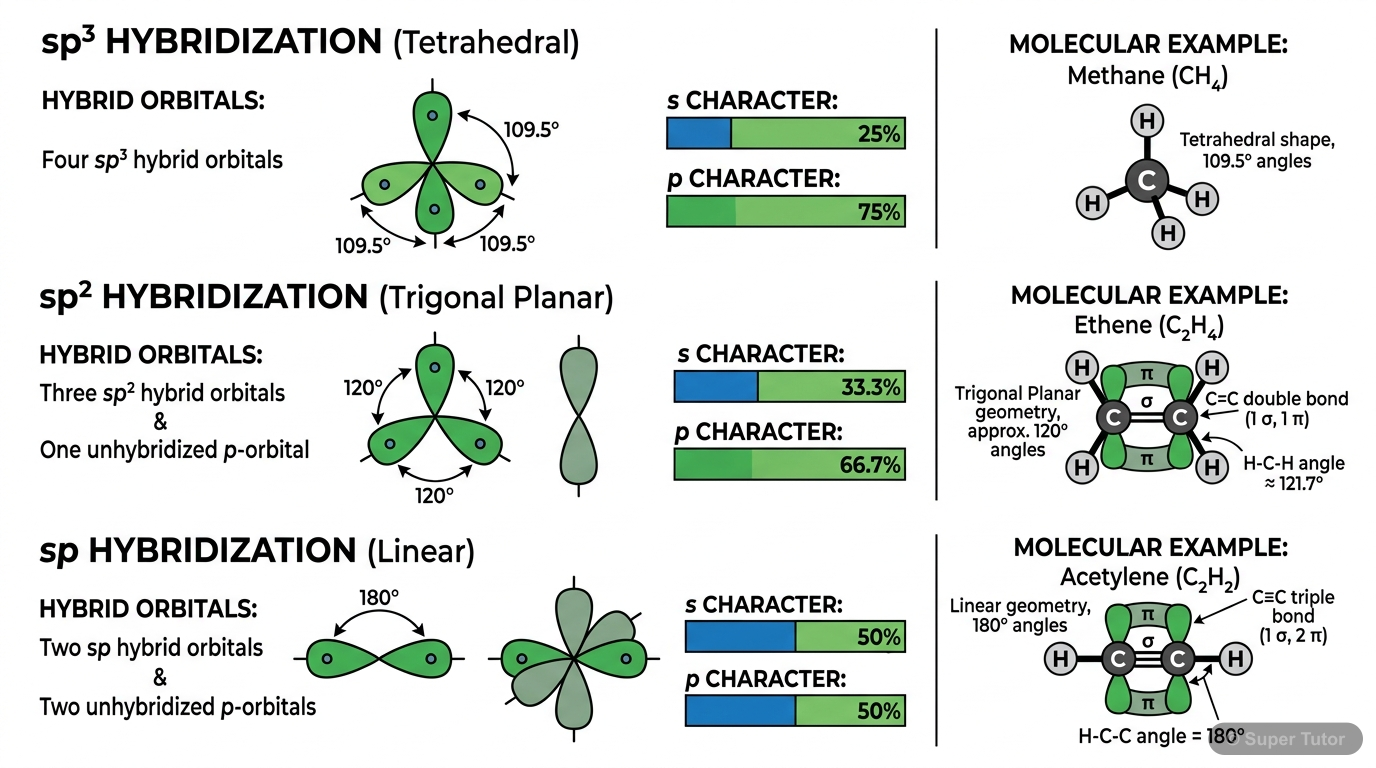
This is just one of 19+ visuals inside Super Tutor's Chemical Bonding and Molecular Structure chapter
Explore the full setRevision Notes — Chemical Bonding and Molecular Structure
Key concepts, formulas, and definitions from Chemical Bonding and Molecular Structure for JEE Advanced Chemistry preparation.
Key Topics to Revise
Kossel-Lewis Approach and Octet Rule
- Kossel explained ionic bond formation through electron transfer to achieve noble gas configuration
- Lewis explained covalent bond formation through electron sharing
- Octet rule: Atoms tend to achieve 8 electrons in their valence shell for stability
Ionic or Electrovalent Bonding
- Formed by complete transfer of electrons from metal to non-metal
- Results in formation of cations and anions held together by electrostatic forces
- Favorable conditions: Low ionization energy of metal, high electron affinity of non-metal, high lattice energy
Covalent Bonding
- Formed by sharing of electron pairs between atoms
- Can be single (σ), double (σ + π), or triple (σ + 2π) bonds
- Directional nature leads to definite molecular shapes
Coordinate or Dative Bonding
- Formed when both electrons in shared pair come from one atom (donor)
- Donor atom has lone pair, acceptor atom has vacant orbital
- Common in complex compounds and coordination complexes
Get complete revision notes with diagrams and examples — continue in Super Tutor
Key Concepts
Frequently Asked Questions
What topics are covered in Chemical Bonding and Molecular Structure for JEE Advanced?
How important is Chemical Bonding and Molecular Structure for JEE Advanced?
How to prepare Chemical Bonding and Molecular Structure for JEE Advanced?
More resources for Chemical Bonding and Molecular Structure
Syllabus
Chemical Bonding and Molecular Structure — syllabus
Important Topics
Chemical Bonding and Molecular Structure — important topics
Practice Questions
Chemical Bonding and Molecular Structure — practice questions
Study Plan
Chemical Bonding and Molecular Structure — study plan
Formula Sheet
Chemical Bonding and Molecular Structure — formula sheet
For JEE Advanced aspirants
Get the full Chemical Bonding and Molecular Structure chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for JEE Advanced Chemistry.