s-Block Group 2 Elements: Alkaline Earth Metals — Revision Notes
JEE Mains · Chemistry
Quick revision notes for s-Block Group 2 Elements: Alkaline Earth Metals — key concepts, formulas, and definitions for JEE Mains Chemistry preparation.
Interactive on Super Tutor
Studying s-Block Group 2 Elements: Alkaline Earth Metals? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for JEE Mains Chemistry.
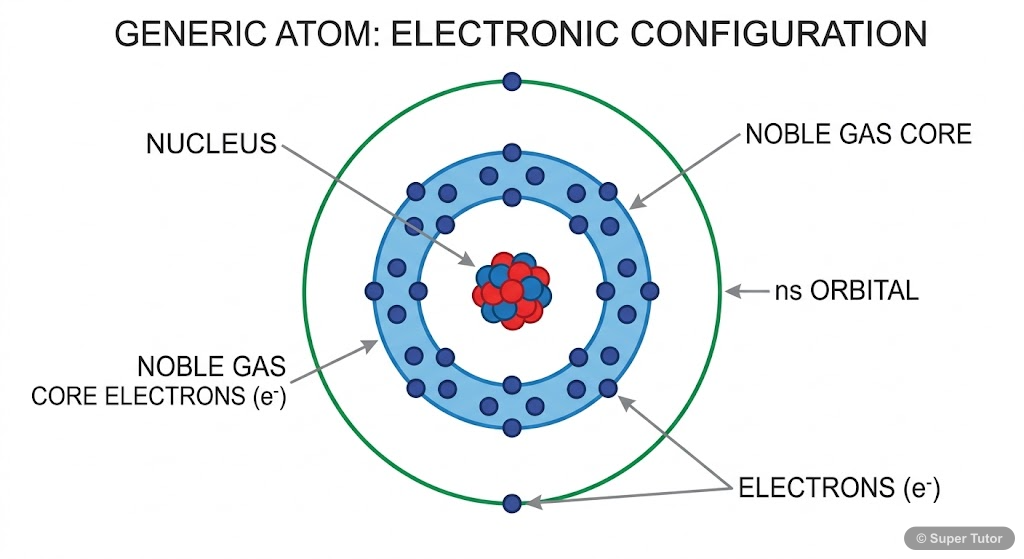
Learn better with visuals Super Tutor has hundreds of illustrations like this across every chapter — all free to try.
Get startedRevision Notes — s-Block Group 2 Elements: Alkaline Earth Metals
Key concepts, formulas, and definitions from s-Block Group 2 Elements: Alkaline Earth Metals for JEE Mains Chemistry preparation.
Key Topics to Revise
General Characteristics and Electronic Configuration
- General electronic configuration: ns² where n = 2 to 7
- All form M²⁺ ions by losing two valence electrons
- Atomic and ionic radii increase down the group: Be < Mg < Ca < Sr < Ba
Chemical Reactivity and Reactions
- Reactivity increases down the group: Be < Mg < Ca < Sr < Ba
- Be doesn't react with water; Mg reacts with hot water; others react with cold water
- All form hydrides (MH₂), oxides (MO), and halides (MX₂)
Important Compounds - Oxides and Hydroxides
- All oxides have rock-salt structure except BeO (wurtzite structure)
- BeO is amphoteric; others are basic with increasing basicity down the group
- Thermal stability of carbonates increases down the group
Halides and Salt-like Compounds
- Be halides are covalent and polymeric; others are ionic
- BeCl₂ has bridged structure in solid state, linear in vapor state
- Hydration tendency decreases down the group
Get complete revision notes with diagrams and examples — continue in Super Tutor
Key Concepts
Frequently Asked Questions
What topics are covered in s-Block Group 2 Elements: Alkaline Earth Metals for JEE Mains?
How important is s-Block Group 2 Elements: Alkaline Earth Metals for JEE Mains?
How to prepare s-Block Group 2 Elements: Alkaline Earth Metals for JEE Mains?
More resources for s-Block Group 2 Elements: Alkaline Earth Metals
Syllabus
s-Block Group 2 Elements: Alkaline Earth Metals — syllabus
Important Topics
s-Block Group 2 Elements: Alkaline Earth Metals — important topics
Practice Questions
s-Block Group 2 Elements: Alkaline Earth Metals — practice questions
Study Plan
s-Block Group 2 Elements: Alkaline Earth Metals — study plan
Formula Sheet
s-Block Group 2 Elements: Alkaline Earth Metals — formula sheet
For JEE Mains aspirants
Get the full s-Block Group 2 Elements: Alkaline Earth Metals chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for JEE Mains Chemistry.