Chapter 23 of 36
Revision Notes
Laws of Thermodynamics — Revision Notes
JEE Mains · Physics
Quick revision notes for Laws of Thermodynamics — key concepts, formulas, and definitions for JEE Mains Physics preparation.
Interactive on Super Tutor
Studying Laws of Thermodynamics? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for JEE Mains Physics.
Super Tutor
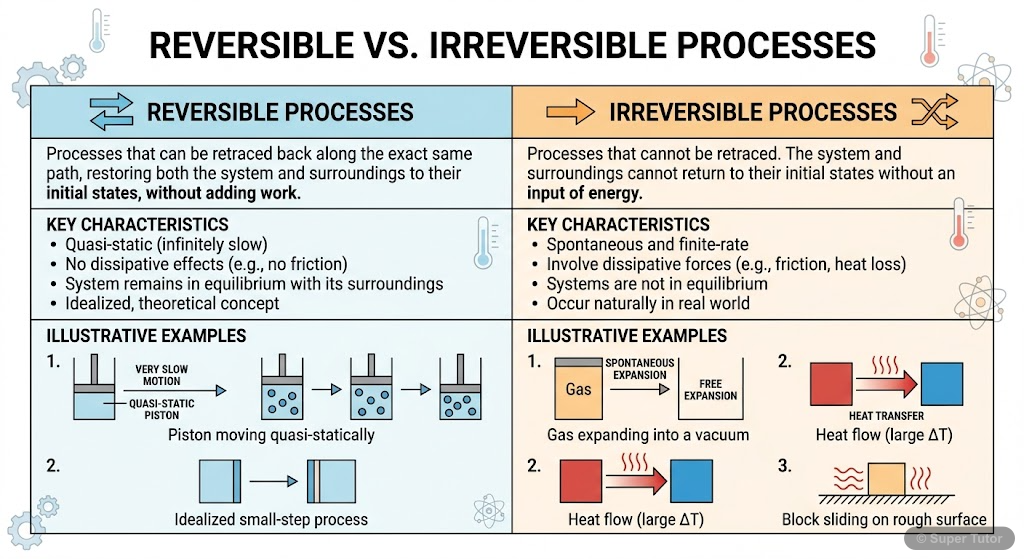
Learn better with visuals Super Tutor has hundreds of illustrations like this across every chapter — all free to try.
Get startedRevision Notes — Laws of Thermodynamics
Key concepts, formulas, and definitions from Laws of Thermodynamics for JEE Mains Physics preparation.
Key Topics to Revise
1
First Law of Thermodynamics
- First law is the principle of conservation of energy applied to thermodynamic systems
- Mathematical expression: Q = W + ΔU where Q = heat supplied, W = work done by system, ΔU = change in internal energy
- Sign conventions: Q > 0 (heat given to system), W > 0 (work done by system), ΔU > 0 (internal energy increases)
2
Thermodynamic Processes
- Isothermal Process: T = constant, pV = constant, ΔU = 0, Q = W
- Isochoric Process: V = constant, W = 0, Q = ΔU = nC_V ΔT
- Isobaric Process: p = constant, W = pΔV, Q = nC_P ΔT
3
Heat Engines and Efficiency
- Heat engine converts thermal energy to mechanical work using working substance
- Components: Hot reservoir (T₁), Cold reservoir (T₂), Working substance
- Energy conservation: Q₁ = W + Q₂ where Q₁ = heat absorbed, Q₂ = heat rejected
4
Refrigerators and Heat Pumps
- Refrigerator transfers heat from cold to hot reservoir using external work
- Operates as reverse heat engine with work input instead of output
- Coefficient of Performance (COP): β = Q₂/W = Q₂/(Q₁-Q₂)
Get complete revision notes with diagrams and examples — continue in Super Tutor
Key Concepts
The first law states that energyFour main typesA heat engine converts heat energyStates that entropy of an isolatedRefrigerators transfer heat from cold
Frequently Asked Questions
What topics are covered in Laws of Thermodynamics for JEE Mains?
Laws of Thermodynamics is an important chapter in JEE Mains Physics. It covers key concepts and formulas that are frequently tested in the exam. Key topics include: First Law of Thermodynamics, Thermodynamic Processes, Heat Engines and Efficiency, Refrigerators and Heat Pumps.
How important is Laws of Thermodynamics for JEE Mains?
Laws of Thermodynamics is a frequently tested chapter in JEE Mains Physics. Questions from this chapter appear regularly in previous year papers. There are 81 practice questions available for this chapter.
How to prepare Laws of Thermodynamics for JEE Mains?
Start by understanding the core concepts, then solve practice questions. Focus on formulas and their applications. Use revision notes for quick review before the exam.
More resources for Laws of Thermodynamics
For JEE Mains aspirants
Get the full Laws of Thermodynamics chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for JEE Mains Physics.