Chapter 32 of 46
Syllabus
Electrochemistry — Syllabus
JEE Mains · Chemistry
Topics covered in Electrochemistry for JEE Mains Chemistry. Understand the syllabus structure and key areas to focus on.
Interactive on Super Tutor
Studying Electrochemistry? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for JEE Mains Chemistry.
Super Tutor
This is just one of 13+ visuals inside Super Tutor's Electrochemistry chapter
Explore the full setElectrochemistry — Syllabus & Topics
Topics covered in Electrochemistry for JEE Mains Chemistry.
Topics in Electrochemistry
1
Electrochemical Cells and EMF
- Galvanic/Voltaic cells convert chemical energy to electrical energy spontaneously
- Electrolytic cells use electrical energy to drive non-spontaneous chemical reactions
- Daniell cell: Zn(s) + Cu²⁺(aq) → Zn²⁺(aq) + Cu(s), E°cell = 1.1 V
2
Standard Electrode Potentials and Electrochemical Series
- Standard Hydrogen Electrode (SHE): 2H⁺ + 2e⁻ → H₂, E° = 0.00 V at 298 K
- Electrochemical series arranges elements by increasing reduction potential
- Higher reduction potential = stronger oxidizing agent, weaker reducing agent
3
Nernst Equation
- Relates cell EMF to concentration of reactants and products
- At equilibrium, Ecell = 0 and reaction quotient equals Keq
- Temperature dependence of cell EMF can be calculated
4
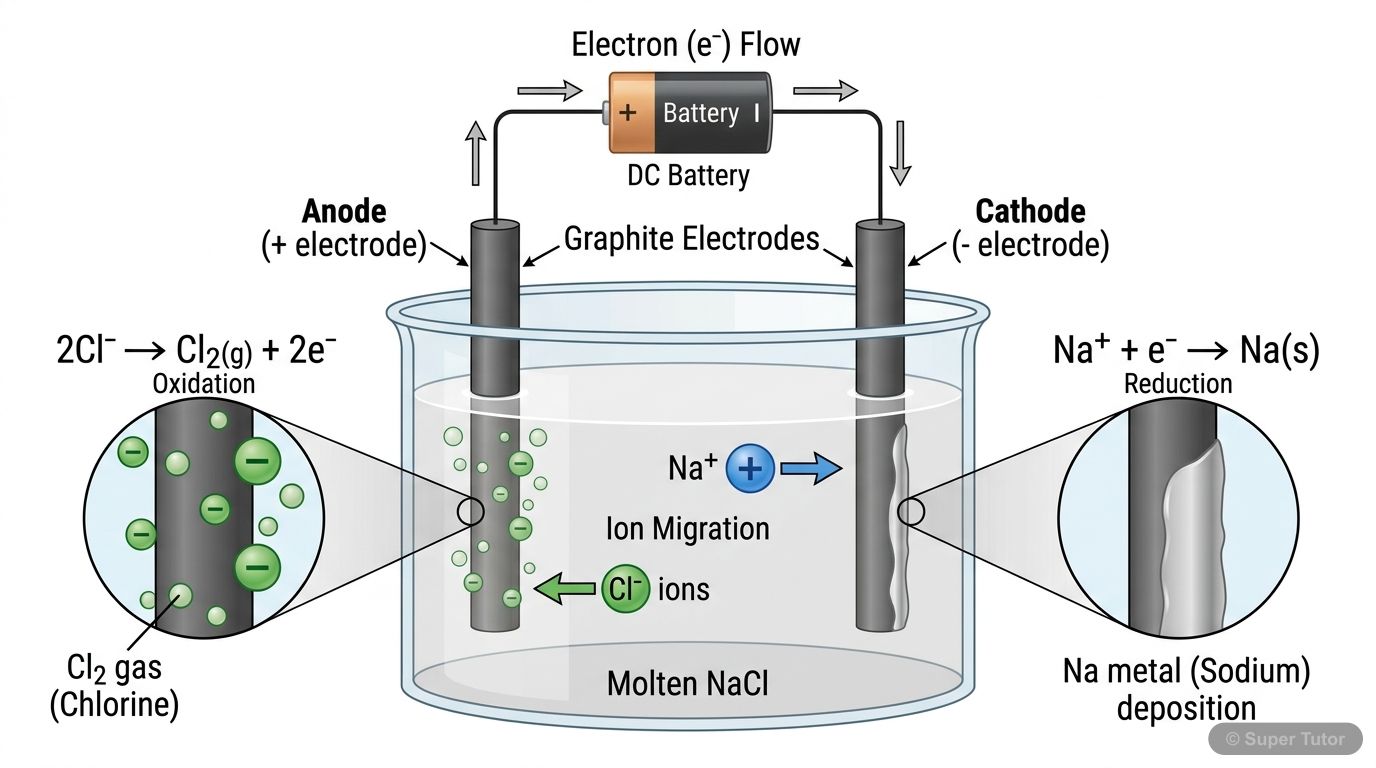
Electrolysis and Faraday's Laws
- Electrolysis uses electrical energy to drive non-spontaneous reactions
- Products depend on electrode potentials, concentration, and overpotential
- Faraday's First Law: Amount deposited ∝ Quantity of electricity passed
Key Concepts
A device that converts chemical energyThe potential difference developed betweenRelates cell potential to concentrationProcess of using electrical energyAbility of electrolytic solutions to conduct
Frequently Asked Questions
What topics are covered in Electrochemistry for JEE Mains?
Electrochemistry is an important chapter in JEE Mains Chemistry. It covers key concepts and formulas that are frequently tested in the exam. Key topics include: Electrochemical Cells and EMF, Standard Electrode Potentials and Electrochemical Series, Nernst Equation, Electrolysis and Faraday's Laws.
How important is Electrochemistry for JEE Mains?
Electrochemistry is a frequently tested chapter in JEE Mains Chemistry. Questions from this chapter appear regularly in previous year papers. There are 90 practice questions available for this chapter.
How to prepare Electrochemistry for JEE Mains?
Start by understanding the core concepts, then solve practice questions. Focus on formulas and their applications. Use revision notes for quick review before the exam.
More resources for Electrochemistry
For JEE Mains aspirants
Get the full Electrochemistry chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for JEE Mains Chemistry.