Electrochemistry — Study Plan
KEAM · Chemistry
Step-by-step study plan for Electrochemistry — structured approach to mastering this chapter for KEAM Chemistry.
Interactive on Super Tutor
Studying Electrochemistry? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for KEAM Chemistry.
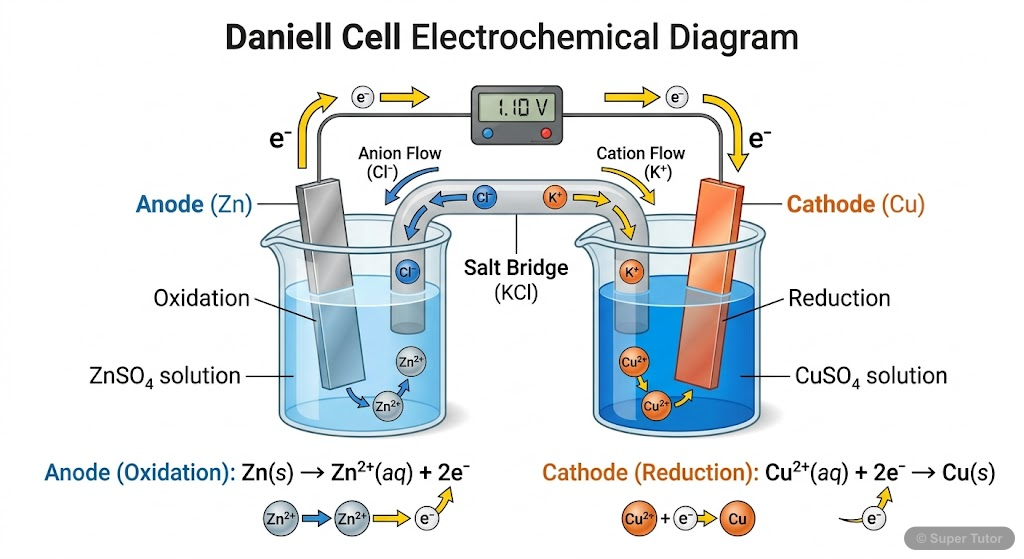
This is just one of 17+ visuals inside Super Tutor's Electrochemistry chapter
Explore the full setHow to Study Electrochemistry
A structured approach to studying Electrochemistry for KEAM Chemistry.
Study Plan for Electrochemistry
Day 1–2: Learn the Theory
Study the chapter thoroughly. Note down definitions, formulas, and key concepts.
Day 3: Practice Problems
Solve practice questions and previous year KEAM problems. There are 271 questions available for this chapter.
Day 4: Revise & Test
Revise key formulas and concepts without looking at notes. Take a practice quiz to test your understanding.
What to Focus On
- Electrochemical cells have two half-cells connected by salt bridge
- Anode: oxidation occurs (negative terminal in galvanic cell)
- Cathode: reduction occurs (positive terminal in galvanic cell)
- Standard conditions: 298K, 1 atm pressure, 1M concentration
- SHE is reference electrode with E° = 0.00V
- Positive E° means easier reduction than H⁺/H₂
- Higher E° = stronger oxidizing agent
- Lower E° = stronger reducing agent
- Nernst equation shows concentration dependence
Common Mistakes to Avoid
In a galvanic cell, the anode is positive and cathode is negative because current flows from positive to negative
Cell potential (EMF) can be calculated as Eanode - Ecathode
In electrolytic cells, the anode is positive and cathode is negative, opposite to galvanic cells
Want a personalised study plan?
Super Tutor creates a day-by-day plan for KEAM Chemistry that adapts to your exam date and pace.
Create My Study Plan — FreeFrequently Asked Questions
What topics are covered in Electrochemistry for KEAM?
How important is Electrochemistry for KEAM?
How to prepare Electrochemistry for KEAM?
More resources for Electrochemistry
For KEAM aspirants
Get the full Electrochemistry chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for KEAM Chemistry.