Organic Chemistry Some Basic Principles and Techniques — Study Plan
MET · Chemistry
Step-by-step study plan for Organic Chemistry Some Basic Principles and Techniques — structured approach to mastering this chapter for MET Chemistry.
Interactive on Super Tutor
Studying Organic Chemistry Some Basic Principles and Techniques? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for MET Chemistry.
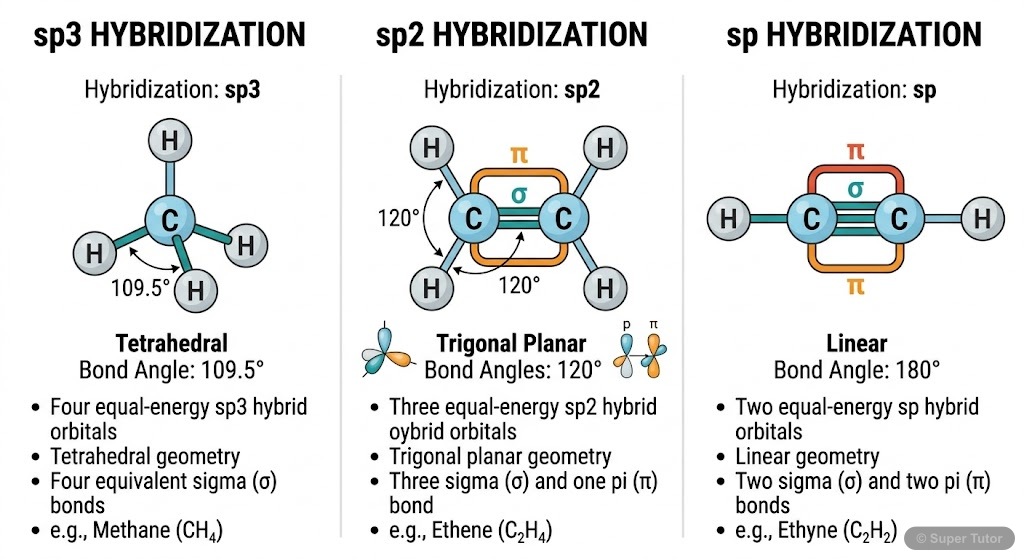
This is just one of 23+ visuals inside Super Tutor's Organic Chemistry Some Basic Principles and Techniques chapter
Explore the full setHow to Study Organic Chemistry Some Basic Principles and Techniques
A structured approach to studying Organic Chemistry Some Basic Principles and Techniques for MET Chemistry.
Study Plan for Organic Chemistry Some Basic Principles and Techniques
Day 1–2: Learn the Theory
Study the chapter thoroughly. Note down definitions, formulas, and key concepts.
Day 3: Practice Problems
Solve practice questions and previous year MET problems. There are 276 questions available for this chapter.
Day 4: Revise & Test
Revise key formulas and concepts without looking at notes. Take a practice quiz to test your understanding.
What to Focus On
- Organic chemistry deals with carbon compounds and their derivatives
- Vital force theory was disproved by Wöhler's synthesis of urea in 1828
- Organic compounds can be synthesized from inorganic sources
- Carbon shows tetravalency through sp³, sp², and sp hybridization
- Hybridization determines molecular geometry and bond angles
- π bonds are weaker than σ bonds and restrict molecular rotation
- Multiple ways exist to represent organic molecule structures
- Condensed formulas group atoms and omit some bonds for simplicity
- Bond-line formulas show carbon framework efficiently
Common Mistakes to Avoid
All carbon atoms in organic compounds are sp3 hybridized and form tetrahedral structures
Carbocations and carbanions have the same stability order
Inductive effect and resonance effect work in the same direction for all groups
Want a personalised study plan?
Super Tutor creates a day-by-day plan for MET Chemistry that adapts to your exam date and pace.
Create My Study Plan — FreeFrequently Asked Questions
What topics are covered in Organic Chemistry Some Basic Principles and Techniques for MET?
How important is Organic Chemistry Some Basic Principles and Techniques for MET?
How to prepare Organic Chemistry Some Basic Principles and Techniques for MET?
More resources for Organic Chemistry Some Basic Principles and Techniques
Syllabus
Organic Chemistry Some Basic Principles and Techniques — syllabus
Revision Notes
Organic Chemistry Some Basic Principles and Techniques — revision notes
Important Topics
Organic Chemistry Some Basic Principles and Techniques — important topics
Practice Questions
Organic Chemistry Some Basic Principles and Techniques — practice questions
Formula Sheet
Organic Chemistry Some Basic Principles and Techniques — formula sheet
For MET aspirants
Get the full Organic Chemistry Some Basic Principles and Techniques chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for MET Chemistry.