Structure Of Atom — Study Plan
MHT-CET · Chemistry
Step-by-step study plan for Structure Of Atom — structured approach to mastering this chapter for MHT-CET Chemistry.
Interactive on Super Tutor
Studying Structure Of Atom? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for MHT-CET Chemistry.
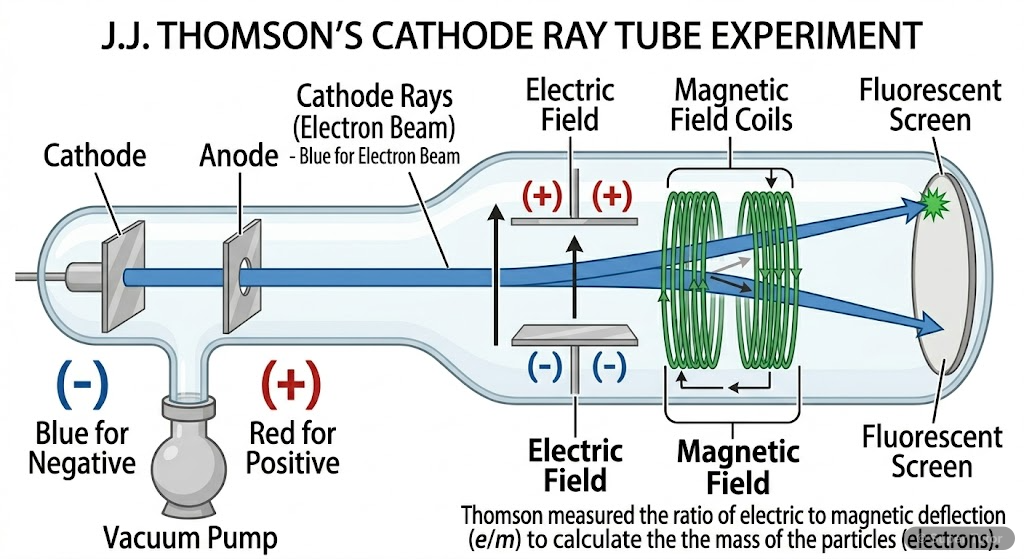
Learn better with visuals Super Tutor has hundreds of illustrations like this across every chapter — all free to try.
Get startedHow to Study Structure Of Atom
A structured approach to studying Structure Of Atom for MHT-CET Chemistry.
Study Plan for Structure Of Atom
Day 1–2: Learn the Theory
Study the chapter thoroughly. Note down definitions, formulas, and key concepts.
Day 3: Practice Problems
Solve practice questions and previous year MHT-CET problems. There are 253 questions available for this chapter.
Day 4: Revise & Test
Revise key formulas and concepts without looking at notes. Take a practice quiz to test your understanding.
What to Focus On
- Cathode rays consist of negatively charged electrons
- Thomson's e/m ratio = 1.758820 × 10¹¹ C kg⁻¹
- Millikan determined electron charge = -1.6 × 10⁻¹⁹ C
- Thomson model: electrons embedded in positive sphere
- Rutherford's experiment proved nuclear model
- Most atomic space is empty
- c = νλ relationship for electromagnetic waves
- Energy is quantized: E = nhν
- Photoelectric effect proves particle nature of light
Common Mistakes to Avoid
Electrons revolve around the nucleus in fixed circular orbits like planets around the sun
Energy levels are equally spaced - the energy difference between n=1 to n=2 is the same as n=2 to n=3
Quantum numbers can have any values as long as they are integers
Want a personalised study plan?
Super Tutor creates a day-by-day plan for MHT-CET Chemistry that adapts to your exam date and pace.
Create My Study Plan — FreeFrequently Asked Questions
What topics are covered in Structure Of Atom for MHT-CET?
How important is Structure Of Atom for MHT-CET?
How to prepare Structure Of Atom for MHT-CET?
More resources for Structure Of Atom
For MHT-CET aspirants
Get the full Structure Of Atom chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for MHT-CET Chemistry.