The D And F Block Elements — Study Plan
OJEE · Chemistry
Step-by-step study plan for The D And F Block Elements — structured approach to mastering this chapter for OJEE Chemistry.
Interactive on Super Tutor
Studying The D And F Block Elements? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for OJEE Chemistry.
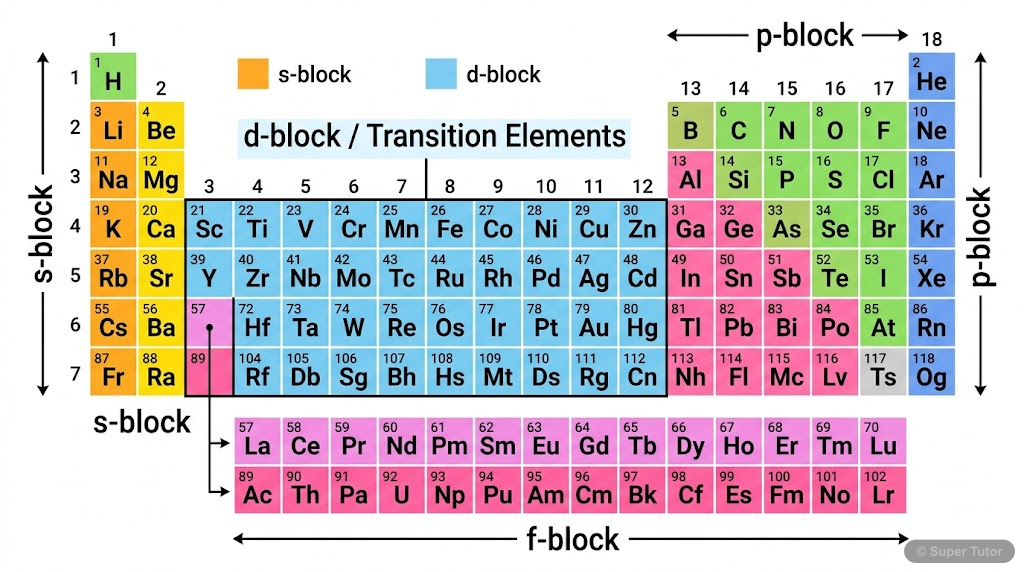
This is just one of 27+ visuals inside Super Tutor's The D And F Block Elements chapter
Explore the full setHow to Study The D And F Block Elements
A structured approach to studying The D And F Block Elements for OJEE Chemistry.
Study Plan for The D And F Block Elements
Day 1–2: Learn the Theory
Study the chapter thoroughly. Note down definitions, formulas, and key concepts.
Day 3: Practice Problems
Solve practice questions and previous year OJEE problems. There are 474 questions available for this chapter.
Day 4: Revise & Test
Revise key formulas and concepts without looking at notes. Take a practice quiz to test your understanding.
What to Focus On
- d-block: (n-1)d¹⁻¹⁰ns⁰⁻² configuration
- f-block: 4f¹⁻¹⁴5d⁰⁻¹6s² (lanthanoids) and 5f¹⁻¹⁴6d⁰⁻²7s² (actinoids)
- Zn, Cd, Hg are not considered transition elements due to completely filled d-orbitals
- High melting/boiling points due to strong metallic bonding
- Atomic radii decrease across period (lanthanoid contraction effect)
- Variable oxidation states due to small energy difference between (n-1)d and ns orbitals
- Color due to d-d transitions in crystal field
- d⁰ and d¹⁰ configurations are colorless
- Paramagnetic behavior due to unpaired electrons
Common Mistakes to Avoid
Zn, Cd, and Hg are transition elements because they are in the d-block
Higher oxidation states are always more stable for transition elements
All transition metal compounds are colored due to d-d transitions
Want a personalised study plan?
Super Tutor creates a day-by-day plan for OJEE Chemistry that adapts to your exam date and pace.
Create My Study Plan — FreeFrequently Asked Questions
What topics are covered in The D And F Block Elements for OJEE?
How important is The D And F Block Elements for OJEE?
How to prepare The D And F Block Elements for OJEE?
More resources for The D And F Block Elements
For OJEE aspirants
Get the full The D And F Block Elements chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for OJEE Chemistry.