Chemical Bonding And Molecular Structure — Revision Notes
SRMJEE · Chemistry
Quick revision notes for Chemical Bonding And Molecular Structure — key concepts, formulas, and definitions for SRMJEE Chemistry preparation.
Interactive on Super Tutor
Studying Chemical Bonding And Molecular Structure? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for SRMJEE Chemistry.
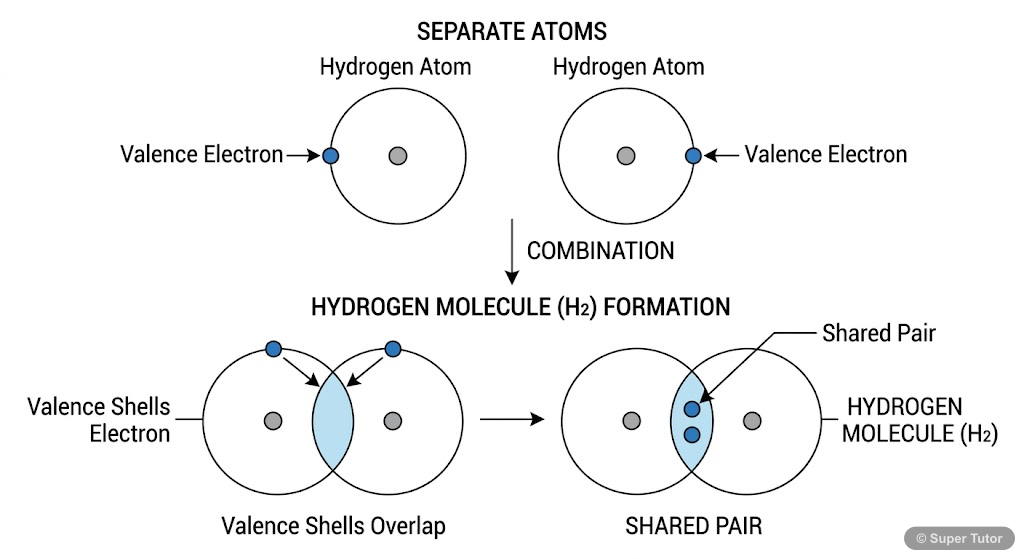
Learn better with visuals Super Tutor has hundreds of illustrations like this across every chapter — all free to try.
Get startedRevision Notes — Chemical Bonding And Molecular Structure
Key concepts, formulas, and definitions from Chemical Bonding And Molecular Structure for SRMJEE Chemistry preparation.
Key Topics to Revise
1. Kössel-Lewis Approach to Chemical Bonding
- Atoms combine to achieve stable noble gas configuration (octet rule)
- Two main ways: electron transfer (ionic bond) or electron sharing (covalent bond)
- Lewis dot structures represent valence electrons as dots around atoms
2. Ionic/Electrovalent Bonding
- Formed by complete transfer of electrons from metal to non-metal
- Results in formation of cations and anions held by electrostatic forces
- Favored when difference in electronegativity ≥ 1.9
3. Covalent Bonding and Bond Parameters
- Formed by sharing of electron pairs between atoms
- Single (σ), double (σ + π), and triple bonds (σ + 2π) possible
- Bond length decreases with increasing bond order
4. VSEPR Theory and Molecular Geometry
- Electron pairs around central atom arrange to minimize repulsion
- Repulsion order: lone pair-lone pair > lone pair-bond pair > bond pair-bond pair
- Both bonding and non-bonding electron pairs determine molecular geometry
Get complete revision notes with diagrams and examples — continue in Super Tutor
Key Concepts
Frequently Asked Questions
What topics are covered in Chemical Bonding And Molecular Structure for SRMJEE?
How important is Chemical Bonding And Molecular Structure for SRMJEE?
How to prepare Chemical Bonding And Molecular Structure for SRMJEE?
More resources for Chemical Bonding And Molecular Structure
Syllabus
Chemical Bonding And Molecular Structure — syllabus
Important Topics
Chemical Bonding And Molecular Structure — important topics
Practice Questions
Chemical Bonding And Molecular Structure — practice questions
Study Plan
Chemical Bonding And Molecular Structure — study plan
Formula Sheet
Chemical Bonding And Molecular Structure — formula sheet
For SRMJEE aspirants
Get the full Chemical Bonding And Molecular Structure chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for SRMJEE Chemistry.