Chapter 3 of 19
Revision Notes
Structure Of Atom — Revision Notes
SRMJEE · Chemistry
Quick revision notes for Structure Of Atom — key concepts, formulas, and definitions for SRMJEE Chemistry preparation.
Interactive on Super Tutor
Studying Structure Of Atom? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for SRMJEE Chemistry.
Super Tutor
Learn better with visuals Super Tutor has hundreds of illustrations like this across every chapter — all free to try.
Get startedRevision Notes — Structure Of Atom
Key concepts, formulas, and definitions from Structure Of Atom for SRMJEE Chemistry preparation.
Key Topics to Revise
1
Discovery of Sub-atomic Particles
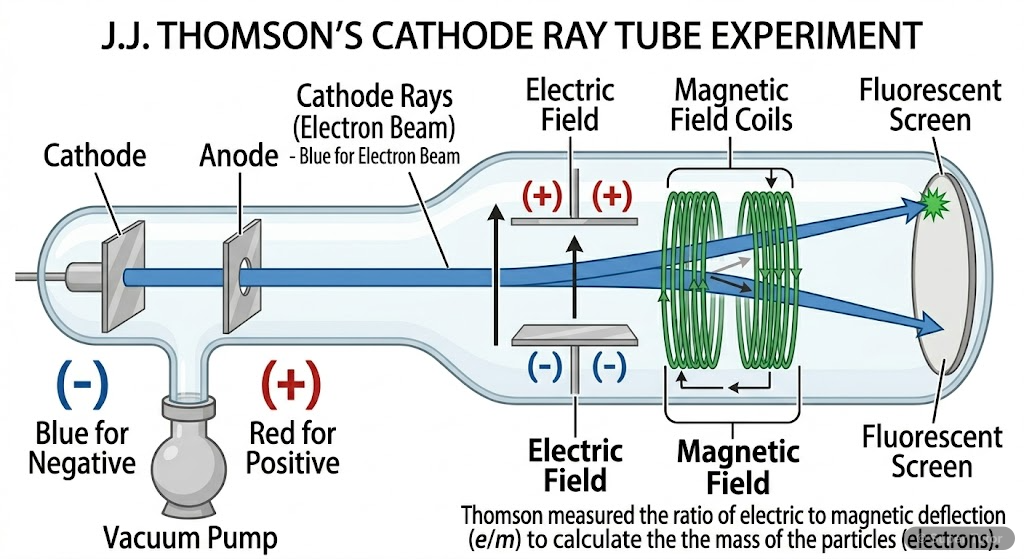
- J.J. Thomson discovered electrons through cathode ray tube experiment in 1897
- Cathode rays consist of negatively charged particles (electrons) that travel from cathode to anode
- Charge to mass ratio of electron: e/me = 1.758820 × 10¹¹ C kg⁻¹
2
Atomic Models
- Thomson's model (1898): 'Raisin pudding' model with electrons embedded in positive sphere
- Rutherford's α-particle scattering experiment revealed nuclear structure
- Most α-particles passed undeflected → atom is mostly empty space
3
Electromagnetic Radiation and Quantum Theory
- Electromagnetic waves have electric and magnetic components perpendicular to each other
- All EM waves travel at speed of light c = 3.0 × 10⁸ m/s in vacuum
- Wave equation: c = νλ where ν = frequency, λ = wavelength
4
Hydrogen Spectrum and Bohr's Model
- Hydrogen shows line spectrum with distinct series: Lyman, Balmer, Paschen, Brackett, Pfund
- Rydberg formula relates wavelengths in hydrogen spectrum
- Bohr's postulates: quantized orbits, no radiation during motion, energy absorbed/emitted during transitions
Get complete revision notes with diagrams and examples — continue in Super Tutor
Key Concepts
JThomson's 'plum pudding' model proposed atomsElectromagnetic radiation travels as wavesBohr proposed electrons revolve in fixedde Broglie proposed matter waves
Frequently Asked Questions
What topics are covered in Structure Of Atom for SRMJEE?
Structure Of Atom is an important chapter in SRMJEE Chemistry. It covers key concepts and formulas that are frequently tested in the exam. Key topics include: Discovery of Sub-atomic Particles, Atomic Models, Electromagnetic Radiation and Quantum Theory, Hydrogen Spectrum and Bohr's Model.
How important is Structure Of Atom for SRMJEE?
Structure Of Atom is a frequently tested chapter in SRMJEE Chemistry. Questions from this chapter appear regularly in previous year papers. There are 253 practice questions available for this chapter.
How to prepare Structure Of Atom for SRMJEE?
Start by understanding the core concepts, then solve practice questions. Focus on formulas and their applications. Use revision notes for quick review before the exam.
More resources for Structure Of Atom
For SRMJEE aspirants
Get the full Structure Of Atom chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for SRMJEE Chemistry.