Chemical Bonding And Molecular Structure — Study Plan
SRMJEE · Chemistry
Step-by-step study plan for Chemical Bonding And Molecular Structure — structured approach to mastering this chapter for SRMJEE Chemistry.
Interactive on Super Tutor
Studying Chemical Bonding And Molecular Structure? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for SRMJEE Chemistry.
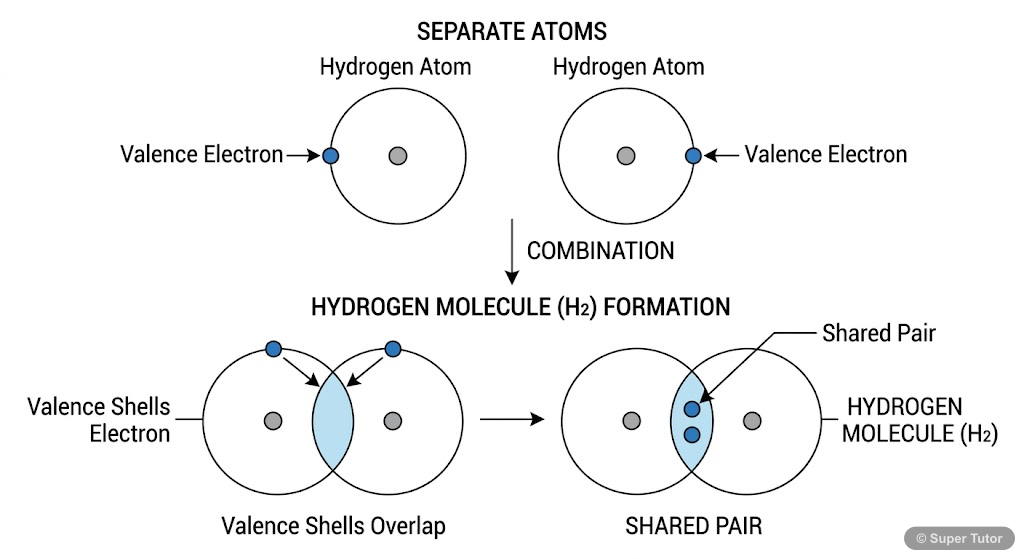
Learn better with visuals Super Tutor has hundreds of illustrations like this across every chapter — all free to try.
Get startedHow to Study Chemical Bonding And Molecular Structure
A structured approach to studying Chemical Bonding And Molecular Structure for SRMJEE Chemistry.
Study Plan for Chemical Bonding And Molecular Structure
Day 1–2: Learn the Theory
Study the chapter thoroughly. Note down definitions, formulas, and key concepts.
Day 3: Practice Problems
Solve practice questions and previous year SRMJEE problems. There are 334 questions available for this chapter.
Day 4: Revise & Test
Revise key formulas and concepts without looking at notes. Take a practice quiz to test your understanding.
What to Focus On
- Chemical bond is the attractive force holding atoms together in molecules
- Atoms interact to achieve noble gas configuration and minimize energy
- Octet rule: atoms need 8 electrons in valence shell for stability
- Ionic bond forms by complete electron transfer between metal and non-metal
- Electrostatic attraction holds cations and anions together
- Lattice enthalpy measures stability of ionic compounds
- Bond length: equilibrium distance between bonded nuclei
- Bond angle: angle between two bonds meeting at an atom
- Bond enthalpy: energy required to break one mole of bonds
Common Mistakes to Avoid
Ionic bonds are formed only between metals and non-metals, and covalent bonds only between non-metals
All atoms must complete their octet to form stable compounds
Lone pairs don't affect molecular geometry - only bonding pairs determine shape
Want a personalised study plan?
Super Tutor creates a day-by-day plan for SRMJEE Chemistry that adapts to your exam date and pace.
Create My Study Plan — FreeFrequently Asked Questions
What topics are covered in Chemical Bonding And Molecular Structure for SRMJEE?
How important is Chemical Bonding And Molecular Structure for SRMJEE?
How to prepare Chemical Bonding And Molecular Structure for SRMJEE?
More resources for Chemical Bonding And Molecular Structure
Syllabus
Chemical Bonding And Molecular Structure — syllabus
Revision Notes
Chemical Bonding And Molecular Structure — revision notes
Important Topics
Chemical Bonding And Molecular Structure — important topics
Practice Questions
Chemical Bonding And Molecular Structure — practice questions
Formula Sheet
Chemical Bonding And Molecular Structure — formula sheet
For SRMJEE aspirants
Get the full Chemical Bonding And Molecular Structure chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for SRMJEE Chemistry.