Chemical Kinetics — Study Plan
TG EAPCET · Chemistry
Step-by-step study plan for Chemical Kinetics — structured approach to mastering this chapter for TG EAPCET Chemistry.
Interactive on Super Tutor
Studying Chemical Kinetics? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for TG EAPCET Chemistry.
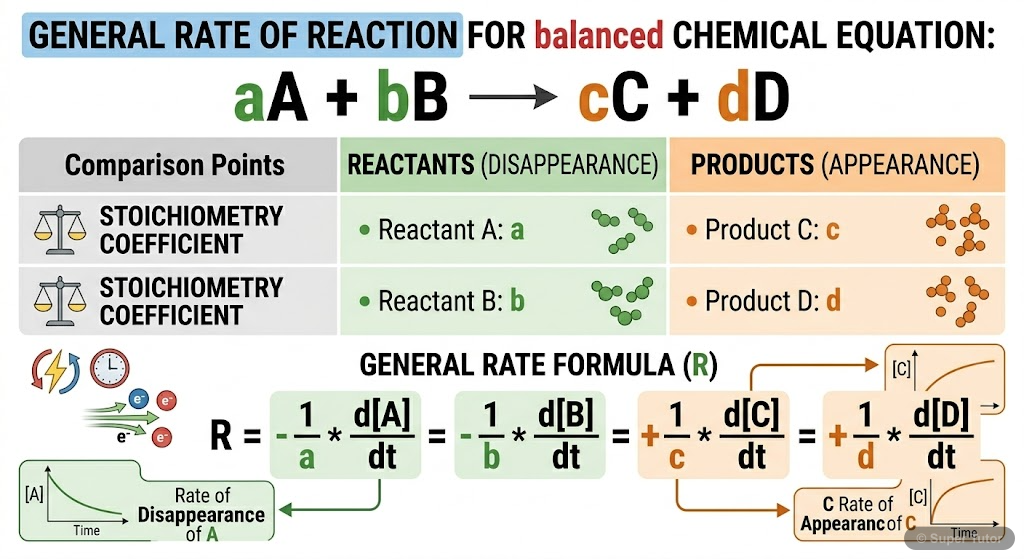
Super Tutor has 16+ illustrations like this for Chemical Kinetics alone — flashcards, concept maps, and step-by-step visuals.
See them allHow to Study Chemical Kinetics
A structured approach to studying Chemical Kinetics for TG EAPCET Chemistry.
Study Plan for Chemical Kinetics
Day 1–2: Learn the Theory
Study the chapter thoroughly. Note down definitions, formulas, and key concepts.
Day 3: Practice Problems
Solve practice questions and previous year TG EAPCET problems. There are 276 questions available for this chapter.
Day 4: Revise & Test
Revise key formulas and concepts without looking at notes. Take a practice quiz to test your understanding.
What to Focus On
- Rate = change in concentration per unit time
- Units: mol L⁻¹ s⁻¹ or atm s⁻¹
- Average rate uses Δ symbol (large changes)
- Higher concentration → faster rate
- Gaseous > liquid > solid reaction rates
- Rate approximately doubles per 10°C rise
- Order = sum of concentration powers in rate law
- Order can be 0, 1, 2, 3, or fractional
- Molecularity = number of molecules in elementary step
Common Mistakes to Avoid
Rate of reaction always depends on stoichiometric coefficients - if the equation is 2A + B → Products, then Rate = k[A]²[B]
Order of reaction is always a whole number (0, 1, 2, 3)
Half-life is always independent of initial concentration for all reactions
Want a personalised study plan?
Super Tutor creates a day-by-day plan for TG EAPCET Chemistry that adapts to your exam date and pace.
Create My Study Plan — FreeFrequently Asked Questions
What topics are covered in Chemical Kinetics for TG EAPCET?
How important is Chemical Kinetics for TG EAPCET?
How to prepare Chemical Kinetics for TG EAPCET?
More resources for Chemical Kinetics
For TG EAPCET aspirants
Get the full Chemical Kinetics chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for TG EAPCET Chemistry.