Chapter 10 of 19
Important Topics
Coordination Compounds — Important Topics
VITEEE · Chemistry
Most important topics from Coordination Compounds for VITEEE Chemistry. Focus on these high-weightage areas for maximum score.
Interactive on Super Tutor
Studying Coordination Compounds? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for VITEEE Chemistry.
Super Tutor
Learn better with visuals Super Tutor has hundreds of illustrations like this across every chapter — all free to try.
Get startedCoordination Compounds — Syllabus & Topics
Topics covered in Coordination Compounds for VITEEE Chemistry.
Topics in Coordination Compounds
1
Werner's Theory of Coordination Compounds
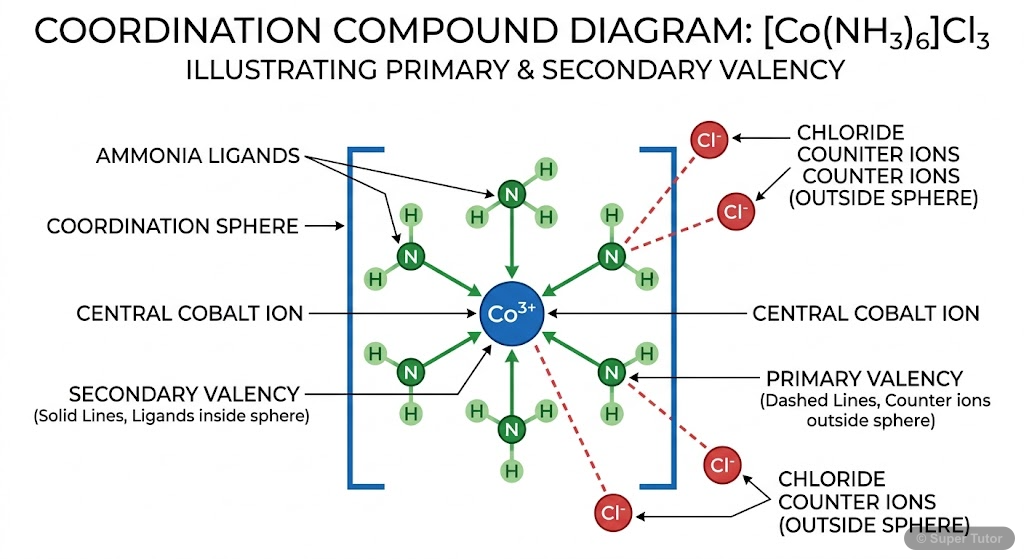
- Alfred Werner proposed this theory in 1898 based on extensive experimental work with cobalt(III) chloride and ammonia complexes
- Metals exhibit two types of valencies: Primary valency (ionizable, corresponds to oxidation state) and Secondary valency (non-ionizable, corresponds to coordination number)
- Primary valency is satisfied by negative ions and is non-directional
2
Important Terminology and Definitions
- Coordination entity/sphere: Central metal atom/ion plus directly attached ligands, written in square brackets
- Counter ions: Ionizable groups written outside square brackets that balance the charge
- Ligands: Ions or molecules that donate electron pairs to the central metal atom through coordinate bonds
3
Nomenclature of Coordination Compounds
- IUPAC rules for writing formulas: Cation first, then anion; coordination sphere in square brackets; ligands in alphabetical order
- Naming rules: Ligands named first (alphabetically), then metal with oxidation state in Roman numerals
- Anionic ligands end in '-ido' (Cl⁻ = chlorido, CN⁻ = cyanido)
4
Isomerism in Coordination Compounds
- Structural isomerism: Different connectivity between atoms (linkage, coordination, ionization, hydration, solvate)
- Stereoisomerism: Same connectivity but different spatial arrangement (geometric and optical)
- Linkage isomerism: Ambidentate ligands coordinate through different donor atoms
Key Concepts
Alfred Werner proposed that metals exhibitLigands are ions or moleculesSystematic naming follows specific rulesStructural isomerism includesMetal provides empty orbitals equal
Frequently Asked Questions
What topics are covered in Coordination Compounds for VITEEE?
Coordination Compounds is an important chapter in VITEEE Chemistry. It covers key concepts and formulas that are frequently tested in the exam. Key topics include: Werner's Theory of Coordination Compounds, Important Terminology and Definitions, Nomenclature of Coordination Compounds, Isomerism in Coordination Compounds.
How important is Coordination Compounds for VITEEE?
Coordination Compounds is a frequently tested chapter in VITEEE Chemistry. Questions from this chapter appear regularly in previous year papers. There are 272 practice questions available for this chapter.
How to prepare Coordination Compounds for VITEEE?
Start by understanding the core concepts, then solve practice questions. Focus on formulas and their applications. Use revision notes for quick review before the exam.
More resources for Coordination Compounds
For VITEEE aspirants
Get the full Coordination Compounds chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for VITEEE Chemistry.