Chapter 11 of 19
Important Topics
Equilibrium — Important Topics
WBJEE · Chemistry
Most important topics from Equilibrium for WBJEE Chemistry. Focus on these high-weightage areas for maximum score.
Interactive on Super Tutor
Studying Equilibrium? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for WBJEE Chemistry.
Super Tutor
This is just one of 27+ visuals inside Super Tutor's Equilibrium chapter
Explore the full setEquilibrium — Syllabus & Topics
Topics covered in Equilibrium for WBJEE Chemistry.
Topics in Equilibrium
1
6.1 Equilibrium in Physical Processes
- Physical equilibrium exists between different phases of the same substance
- Examples: solid ⇌ liquid, liquid ⇌ gas, solid ⇌ gas
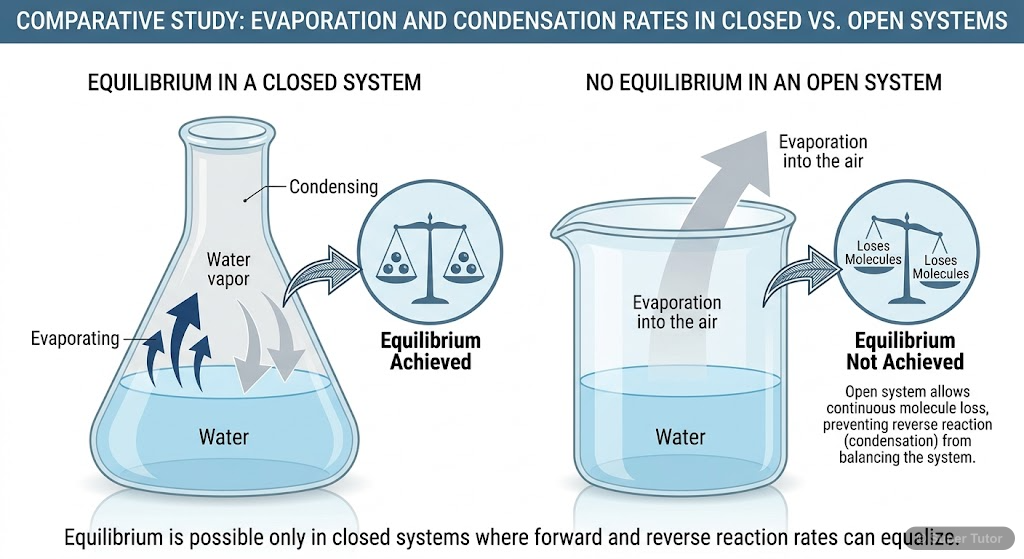
- Equilibrium can only be established in closed systems
2
6.2-6.3 Chemical Equilibrium and Law of Mass Action
- Chemical equilibrium involves reversible chemical reactions
- At equilibrium: rate of forward reaction = rate of backward reaction
- Law of Mass Action: For aA + bB ⇌ cC + dD
3
6.4-6.5 Homogeneous and Heterogeneous Equilibria
- Homogeneous equilibria: all species in same phase
- Heterogeneous equilibria: species in different phases
- For gas phase reactions: Kp = (pC)ᶜ(pD)ᵈ/(pA)ᵃ(pB)ᵇ
4
6.6-6.7 Applications and Thermodynamic Relations
- Reaction quotient Qc predicts reaction direction
- If Qc < Kc: reaction proceeds forward
- If Qc > Kc: reaction proceeds backward
Key Concepts
At equilibriumFor reaction aA + bB ⇌When a system at equilibriumAcids donate protons (H⁺) while basesSolutions that resist pH changes upon
Frequently Asked Questions
What topics are covered in Equilibrium for WBJEE?
Equilibrium is an important chapter in WBJEE Chemistry. It covers key concepts and formulas that are frequently tested in the exam. Key topics include: 6.1 Equilibrium in Physical Processes, 6.2-6.3 Chemical Equilibrium and Law of Mass Action, 6.4-6.5 Homogeneous and Heterogeneous Equilibria, 6.6-6.7 Applications and Thermodynamic Relations.
How important is Equilibrium for WBJEE?
Equilibrium is a frequently tested chapter in WBJEE Chemistry. Questions from this chapter appear regularly in previous year papers. There are 284 practice questions available for this chapter.
How to prepare Equilibrium for WBJEE?
Start by understanding the core concepts, then solve practice questions. Focus on formulas and their applications. Use revision notes for quick review before the exam.
More resources for Equilibrium
For WBJEE aspirants
Get the full Equilibrium chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for WBJEE Chemistry.