Redox Reactions — Study Plan
WBJEE · Chemistry
Step-by-step study plan for Redox Reactions — structured approach to mastering this chapter for WBJEE Chemistry.
Interactive on Super Tutor
Studying Redox Reactions? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for WBJEE Chemistry.
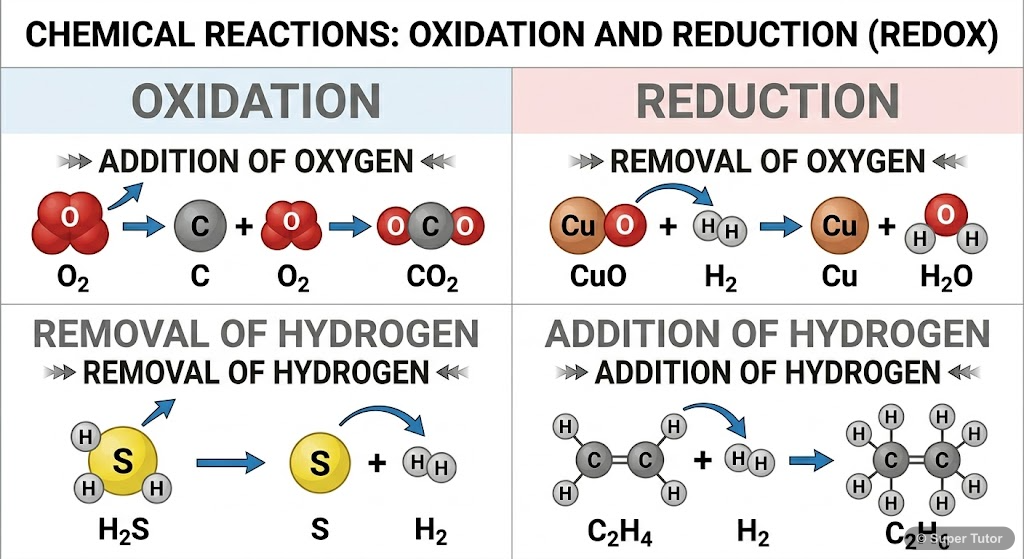
This is just one of 12+ visuals inside Super Tutor's Redox Reactions chapter
Explore the full setHow to Study Redox Reactions
A structured approach to studying Redox Reactions for WBJEE Chemistry.
Study Plan for Redox Reactions
Day 1–2: Learn the Theory
Study the chapter thoroughly. Note down definitions, formulas, and key concepts.
Day 3: Practice Problems
Solve practice questions and previous year WBJEE problems. There are 172 questions available for this chapter.
Day 4: Revise & Test
Revise key formulas and concepts without looking at notes. Take a practice quiz to test your understanding.
What to Focus On
- Classical oxidation involves gain of oxygen or loss of hydrogen
- Classical reduction involves loss of oxygen or gain of hydrogen
- Redox reactions involve simultaneous oxidation and reduction
- Oxidation is loss of electrons (OIL - Oxidation Is Loss)
- Reduction is gain of electrons (RIG - Reduction Is Gain)
- Oxidizing agent gets reduced, reducing agent gets oxidized
- Oxidation number helps identify electron transfer in compounds
- Sum rule: neutral compounds sum to 0, ions sum to their charge
- Oxygen is usually -2, hydrogen usually +1
Common Mistakes to Avoid
Oxidation always means addition of oxygen and reduction always means removal of oxygen
In fractional oxidation states, all atoms of the element have the same fractional oxidation number
The substance that gets oxidized is the oxidizing agent
Want a personalised study plan?
Super Tutor creates a day-by-day plan for WBJEE Chemistry that adapts to your exam date and pace.
Create My Study Plan — FreeFrequently Asked Questions
What topics are covered in Redox Reactions for WBJEE?
How important is Redox Reactions for WBJEE?
How to prepare Redox Reactions for WBJEE?
More resources for Redox Reactions
For WBJEE aspirants
Get the full Redox Reactions chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for WBJEE Chemistry.