Analytical chemistry
ICSE · Class 10 · Chemistry
Flashcards for Analytical chemistry — ICSE Class 10 Chemistry. Quick Q&A cards covering key concepts, definitions, and formulas.
Interactive on Super Tutor
Studying Analytical chemistry? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for flashcards and more.
1,000+ Class 10 students started this chapter today
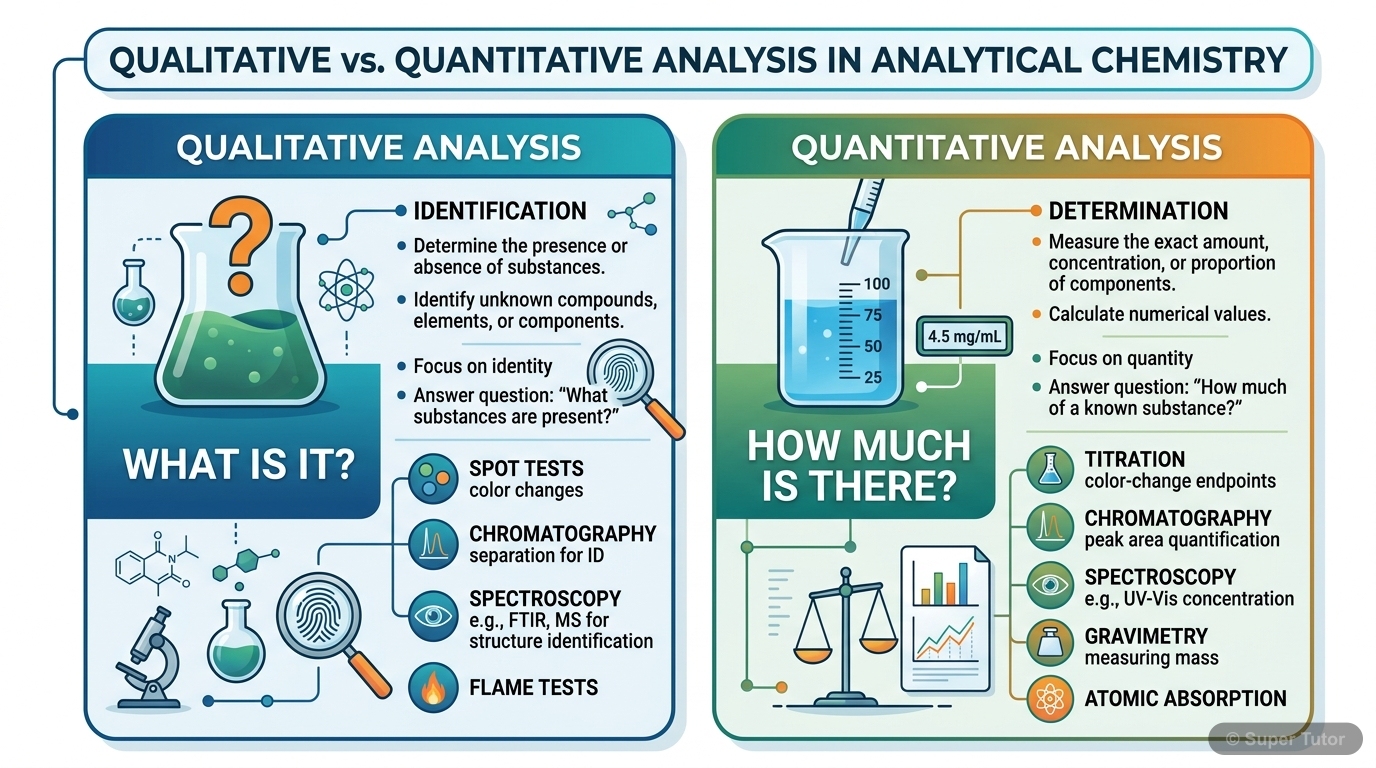
Learn better with visuals Super Tutor has hundreds of illustrations like this across every chapter — all free to try.
Get startedWhat is analytical chemistry?
Answer
Analytical chemistry is the branch of chemistry that deals with the analysis of a chemical substance or its solution to explore its nature, composition and constitution. It includes qualitative analys…
What causes the color of salts and their solutions?
Answer
The color of a salt is due to the presence of colored ions in it. For example, CuSO₄ is blue because it contains blue Cu²⁺ ions and colorless SO₄²⁻ ions. Ions of normal elements (groups IA-VIIA) are c…
Name three colored cations and their colors.
Answer
Cu²⁺ (cupric ion) - Blue Fe²⁺ (ferrous ion) - Light green Fe³⁺ (ferric ion) - Pale yellow…
List five colorless cations commonly found in salts.
Answer
NH₄⁺ (ammonium ion) Na⁺ (sodium ion) K⁺ (potassium ion) Mg²⁺ (magnesium ion) Ca²⁺ (calcium ion) Al³⁺ (aluminum ion) Pb²⁺ (lead ion) Zn²⁺ (zinc ion)…
What happens when NH₄OH is added to a calcium salt solution?
Answer
A dull white precipitate of Ca(OH)₂ is formed which remains insoluble even in excess NH₄OH. Example: CaCl₂ + 2NH₄OH → Ca(OH)₂↓ + 2NH₄Cl…
Describe the action of NH₄OH on ferrous (Fe²⁺) salt solutions.
Answer
A dirty green precipitate of Fe(OH)₂ is formed which is insoluble in excess NH₄OH. Example: FeSO₄ + 2NH₄OH → Fe(OH)₂↓ + (NH₄)₂SO₄ The solution is initially pale green colored.
What precipitate is formed when NH₄OH is added to ferric (Fe³⁺) salt solutions?
Answer
A reddish brown precipitate of Fe(OH)₃ is formed which is insoluble in excess NH₄OH. Example: FeCl₃ + 3NH₄OH → Fe(OH)₃↓ + 3NH₄Cl The original solution is yellow colored.
What is special about the action of NH₄OH on copper(II) salt solutions?
Answer
Initially forms a pale blue precipitate of Cu(OH)₂. In excess NH₄OH, this dissolves to form a deep blue soluble complex [Cu(NH₃)₄]SO₄ (tetraamine copper(II) sulphate). CuSO₄ + 2NH₄OH → Cu(OH)₂↓ + (NH₄…
+12 more flashcards available
Practice AllFrequently Asked Questions
What are the important topics in Analytical chemistry for ICSE Class 10 Chemistry?
How to score full marks in Analytical chemistry — ICSE Class 10 Chemistry?
How many flashcards are available for Analytical chemistry?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Analytical chemistry
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Analytical chemistry chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for ICSE Class 10 Chemistry.