Study of Compounds
ICSE · Class 10 · Chemistry
Flashcards for Study of Compounds — ICSE Class 10 Chemistry. Quick Q&A cards covering key concepts, definitions, and formulas.
Interactive on Super Tutor
Studying Study of Compounds? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for flashcards and more.
1,000+ Class 10 students started this chapter today
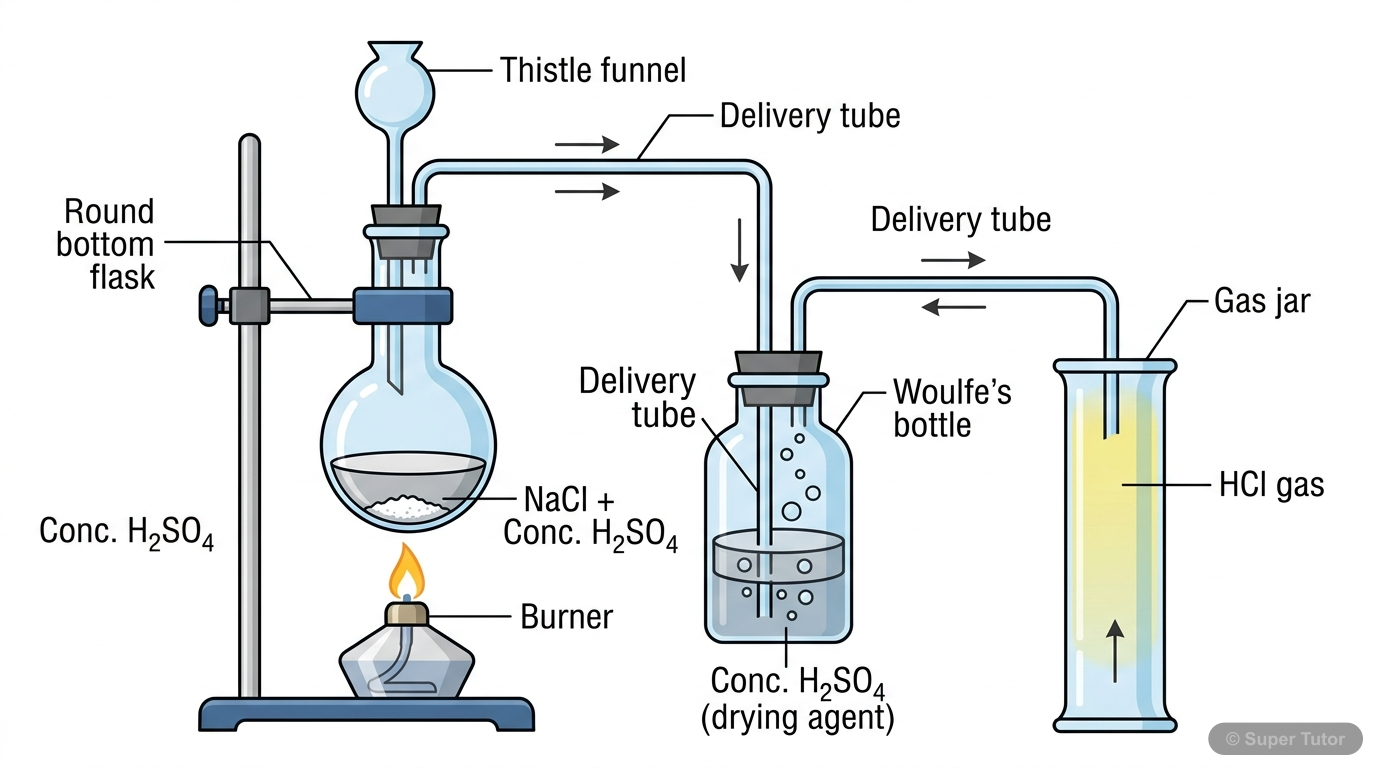
This is just one of 14+ visuals inside Super Tutor's Study of Compounds chapter
Explore the full setWhat is the laboratory method for preparing Hydrogen Chloride gas? Write the chemical equation.
Answer
Laboratory method: Heating sodium chloride with concentrated sulphuric acid at 200°C. Equation: NaCl + H2SO4 → NaHSO4 + HCl(g)↑ Apparatus: Round bottom flask with thistle funnel, delivery tube throu…
Why is HCl gas collected by upward displacement of air and not over water?
Answer
HCl gas is collected by upward displacement of air because: 1. It is 1.27 times heavier than air (VD of HCl = 18.25) 2. It cannot be collected over water because it is highly soluble in water (452 vol…
Describe the fountain experiment for HCl gas and explain the observations.
Answer
Setup: Round bottom flask with dry HCl gas, fitted with jet tube and water dropper, inverted over blue litmus solution. Procedure: Press dropper to add water. Observation: Red fountain forms. Expla…
What is aqua regia? Write its composition and use.
Answer
Aqua regia is a mixture of 1 part conc. HNO3 and 3 parts conc. HCl by volume. Reaction: HNO3 + 3HCl → NOCl + 2H2O + 2[Cl] Use: Dissolves noble metals like gold and platinum due to nascent chlorine: …
Why is inverted funnel arrangement used in laboratory preparation of HCl acid?
Answer
The inverted funnel arrangement prevents back suction. Reason: HCl gas is highly soluble in water, creating partial vacuum when it dissolves. This could cause water to rise up a simple delivery tube …
What is the laboratory preparation of ammonia from ammonium chloride? Write the equation and conditions.
Answer
Reactants: Ammonium chloride (NH4Cl) and calcium hydroxide [Ca(OH)2] in 2:3 ratio by weight. Equation: 2NH4Cl + Ca(OH)2 → CaCl2 + 2H2O + 2NH3↑ Conditions: Heat the mixture Apparatus: Round bottom f…
Why cannot conc. H2SO4, P2O5, and anhydrous CaCl2 be used for drying ammonia gas?
Answer
These drying agents react chemically with ammonia: 1. 2NH3 + H2SO4 → (NH4)2SO4 2. 6NH3 + P2O5 + 3H2O → 2(NH4)3PO4 3. 4NH3 + CaCl2 → CaCl2·4NH3 Instead, quick lime (CaO) is used as it only absorbs mo…
Describe Haber's process for manufacture of ammonia. Include conditions and equation.
Answer
Equation: N2 + 3H2 ⇌ 2NH3 + 93.6 kJ/mol Conditions: - Temperature: 450-500°C (optimum) - Pressure: 200-900 atm - Catalyst: Finely divided iron with molybdenum promoter Process: Compressed N2 and H2 …
+16 more flashcards available
Practice AllFrequently Asked Questions
What are the important topics in Study of Compounds for ICSE Class 10 Chemistry?
How to score full marks in Study of Compounds — ICSE Class 10 Chemistry?
How many flashcards are available for Study of Compounds?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Study of Compounds
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Study of Compounds chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for ICSE Class 10 Chemistry.