Study of Gas Laws
ICSE · Class 9 · Chemistry
Summary of Study of Gas Laws for ICSE Class 9 Chemistry. Key concepts, important points, and chapter overview.
Interactive on Super Tutor
Studying Study of Gas Laws? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for chapter summary and more.
1,000+ Class 9 students started this chapter today
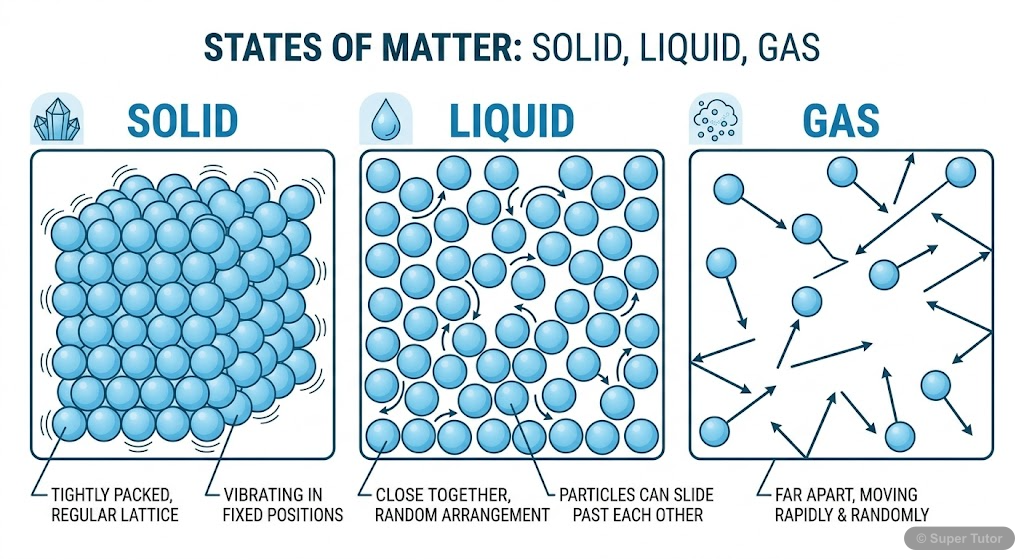
Super Tutor has 12+ illustrations like this for Study of Gas Laws alone — flashcards, concept maps, and step-by-step visuals.
See them allOverview
This chapter explores the fascinating behavior of gases and the scientific laws that govern their properties. Gases are unique among the states of matter because they have neither fixed volume nor shape, and their particles move freely in all directions. Understanding gas laws helps us explain every
Key Concepts
Gases have no fixed shape
Gases have no fixed shape or volume, are highly compressible, have low density, exert pressure in all directions, and can diffuse rapidly. Gas particl
At constant temperature
At constant temperature, the volume of a fixed mass of gas is inversely proportional to its pressure. Mathematically: P₁V₁ = P₂V₂. When pressure doubl
At constant pressure
At constant pressure, the volume of a fixed mass of gas is directly proportional to its absolute temperature. Mathematically: V₁/T₁ = V₂/T₂. Gas expan
Absolute zero (
Absolute zero (-273°C or 0 K) is the theoretical temperature at which all molecular motion ceases. The Kelvin scale starts from absolute zero, making
When both pressure and temperature change
When both pressure and temperature change, the relationship is given by P₁V₁/T₁ = P₂V₂/T₂. This combines both Boyle's and Charles's laws into one comp
Learning Objectives
- Understand the characteristic properties and behavior of gases in terms of molecular motion
- Learn and apply Boyle's Law to solve problems involving pressure-volume relationships
- Master Charles's Law and its applications in temperature-volume calculations
- Understand the concept of absolute zero and the Kelvin temperature scale
- Apply the combined gas equation to solve complex problems involving pressure, volume, and temperature changes
Frequently Asked Questions
What are the important topics in Study of Gas Laws for ICSE Class 9 Chemistry?
How to score full marks in Study of Gas Laws — ICSE Class 9 Chemistry?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Study of Gas Laws
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Flashcards
Quick-fire cards for active recall
Formula Sheet
All formulas in one place
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Study of Gas Laws chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for ICSE Class 9 Chemistry.