Atomic Structure and Chemical Bonding
ICSE · Class 9 · Chemistry
Flashcards for Atomic Structure and Chemical Bonding — ICSE Class 9 Chemistry. Quick Q&A cards covering key concepts, definitions, and formulas.
Interactive on Super Tutor
Studying Atomic Structure and Chemical Bonding? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for flashcards and more.
1,000+ Class 9 students started this chapter today
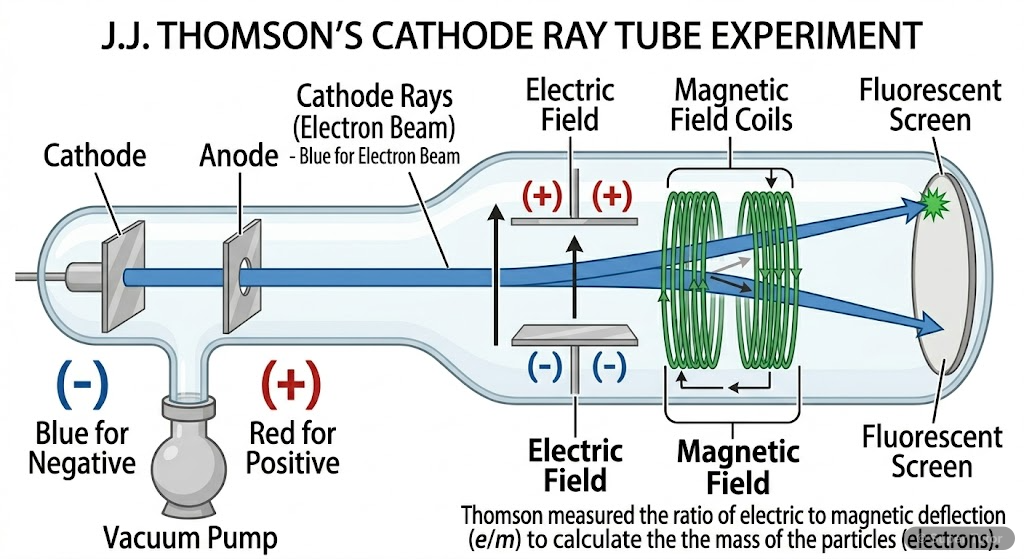
Learn better with visuals Super Tutor has hundreds of illustrations like this across every chapter — all free to try.
Get startedWhat is an atom? Give its definition.
Answer
An atom is the smallest particle of an element that exhibits all the properties of that element. It may or may not exist independently but takes part in every chemical reaction. It is the smallest pos…
Name the three fundamental particles of an atom and their charges.
Answer
The three fundamental particles are: 1. Electron (e⁻) - Negative charge (-1) 2. Proton (p⁺) - Positive charge (+1) 3. Neutron (n) - No charge (0 or neutral)…
What is the atomic number (Z) of an element?
Answer
Atomic number (Z) is the number of protons present in the nucleus of an atom. It is also equal to the number of electrons in a neutral atom. It distinguishes one element from another and gives the tot…
What is mass number (A) and how is it calculated?
Answer
Mass number (A) is the total number of protons and neutrons present in the nucleus of an atom. Formula: A = Number of protons + Number of neutrons Since protons = atomic number (Z), we can write: A = …
State the 2n² rule for electron distribution in shells.
Answer
The 2n² rule states that the maximum number of electrons that can be accommodated in any shell is given by 2n², where n is the shell number. K shell (n=1): 2×1² = 2 electrons L shell (n=2): 2×2² = 8 e…
What are valence electrons and why are they important?
Answer
Valence electrons are the electrons present in the outermost shell (valence shell) of an atom. They are important because: 1. They determine the chemical properties of elements 2. They take part in ch…
State the octet rule and explain its significance.
Answer
The octet rule states that atoms tend to combine with other atoms to attain 8 electrons in their outermost shell (like noble gases). This gives them a stable electronic configuration. Exception: Heliu…
What are isotopes? Give an example.
Answer
Isotopes are atoms of the same element having the same atomic number but different mass numbers (different number of neutrons). Example: Hydrogen isotopes - Protium: ¹₁H (0 neutrons) - Deuterium: ²₁H …
+17 more flashcards available
Practice AllFrequently Asked Questions
What are the important topics in Atomic Structure and Chemical Bonding for ICSE Class 9 Chemistry?
How to score full marks in Atomic Structure and Chemical Bonding — ICSE Class 9 Chemistry?
How many flashcards are available for Atomic Structure and Chemical Bonding?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Atomic Structure and Chemical Bonding
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Atomic Structure and Chemical Bonding chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for ICSE Class 9 Chemistry.