Study of the First Element - Hydrogen
ICSE · Class 9 · Chemistry
Flashcards for Study of the First Element - Hydrogen — ICSE Class 9 Chemistry. Quick Q&A cards covering key concepts, definitions, and formulas.
Interactive on Super Tutor
Studying Study of the First Element - Hydrogen? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for flashcards and more.
1,000+ Class 9 students started this chapter today
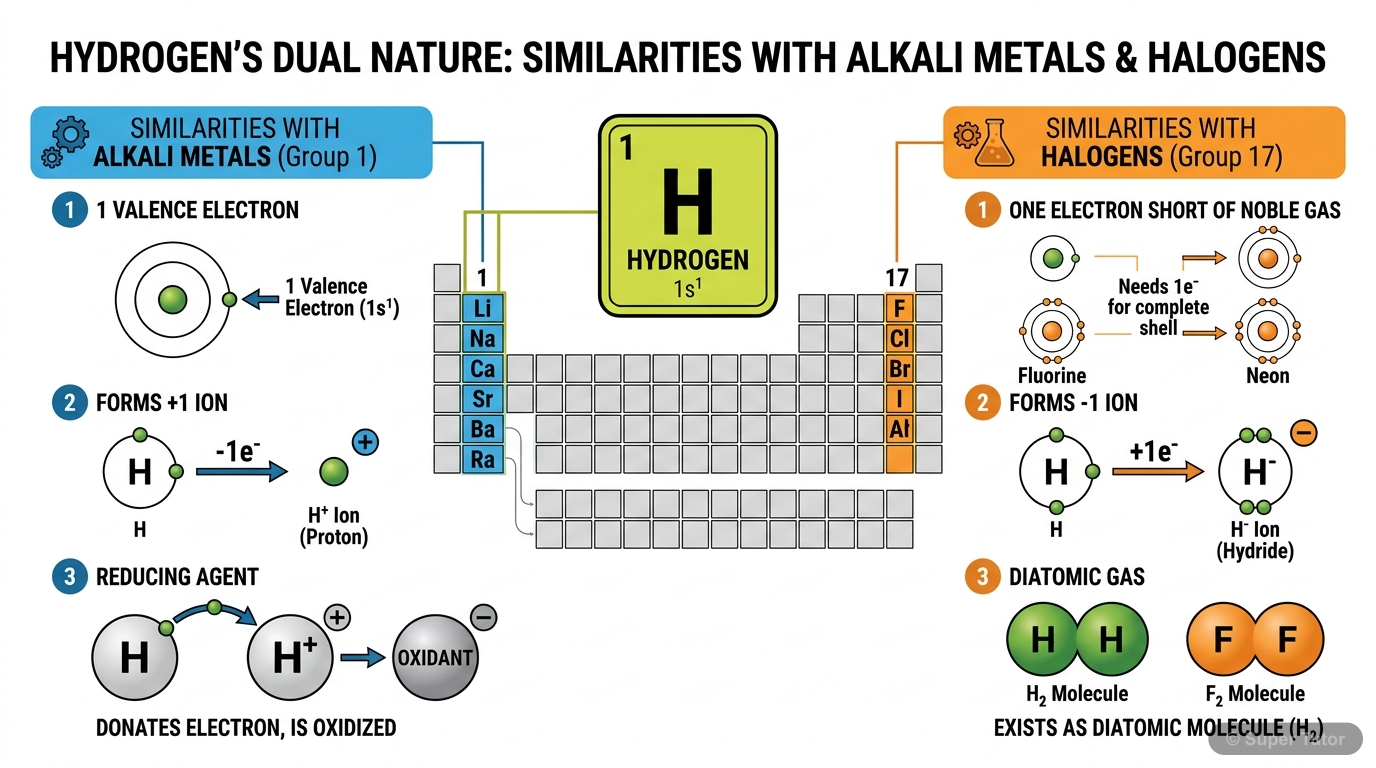
Super Tutor has 11+ illustrations like this for Study of the First Element - Hydrogen alone — flashcards, concept maps, and step-by-step visuals.
See them allWhat is the position of hydrogen in the periodic table and why is it considered unique?
Answer
Hydrogen is placed in the 1st period and Group IA of the periodic table. It is unique because it shows dual nature - it resembles both alkali metals (Group IA) and halogens (Group VIIA) in its propert…
List three similarities between hydrogen and alkali metals.
Answer
1. Both have one electron in their outermost shell 2. Both have valency 1 3. Both can form positive ions by losing electrons (H → H⁺ + e⁻, Na → Na⁺ + e⁻) 4. Both act as reducing agents 5. Both burn in…
How do hydrogen and halogens show similar properties?
Answer
1. Both are one electron short of nearest noble gas configuration 2. Both have valency 1 3. Both can form negative ions (H⁻, Cl⁻) 4. Both are non-metals showing electronegative character 5. Both exist…
Write the balanced equation when sodium reacts with cold water and state your observations.
Answer
Equation: 2Na + 2H₂O → 2NaOH + H₂ Observations: - Sodium floats on water (density 0.97 g/cc) - It melts forming a silvery globule - It darts about on water surface - Burns with golden yellow flame - …
Why does the reaction between potassium and water catch fire?
Answer
The reaction between potassium and water is highly exothermic (releases large amount of heat). The heat generated is sufficient to ignite the hydrogen gas produced, causing the potassium to catch fire…
Write the balanced equation for the reaction between red hot iron and steam. Why is this reaction considered reversible?
Answer
Equation: 3Fe + 4H₂O ⇌ Fe₃O₄ + 4H₂ This reaction is reversible because: - Forward reaction: Iron reacts with steam to form iron oxide and hydrogen - Backward reaction: If hydrogen is not removed, it …
What is the activity series and how does it help in hydrogen preparation?
Answer
Activity series is the arrangement of metals in decreasing order of reactivity: K > Na > Ca > Mg > Al > Zn > Fe > Pb > [H] > Cu > Hg > Ag > Au It helps because: - Metals above hydrogen can displace h…
Which metals show unique nature by reacting with both acids and alkalis to produce hydrogen?
Answer
Zinc, Lead, and Aluminium show unique nature. Examples: Zn + 2HCl → ZnCl₂ + H₂ (with acid) Zn + 2NaOH → Na₂ZnO₂ + H₂ (with alkali) Al + 3HCl → AlCl₃ + 3H₂ (with acid) 2Al + 2NaOH + 2H₂O → 2NaAlO₂ + …
+16 more flashcards available
Practice AllFrequently Asked Questions
What are the important topics in Study of the First Element - Hydrogen for ICSE Class 9 Chemistry?
How to score full marks in Study of the First Element - Hydrogen — ICSE Class 9 Chemistry?
How many flashcards are available for Study of the First Element - Hydrogen?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Study of the First Element - Hydrogen
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Study of the First Element - Hydrogen chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for ICSE Class 9 Chemistry.