Atomic Structure and Chemical Bonding
ICSE · Class 9 · Chemistry
Quick revision notes for Atomic Structure and Chemical Bonding — ICSE Class 9 Chemistry. Key concepts, formulas, and definitions for last-minute revision.
Interactive on Super Tutor
Studying Atomic Structure and Chemical Bonding? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for revision notes and more.
1,000+ Class 9 students started this chapter today
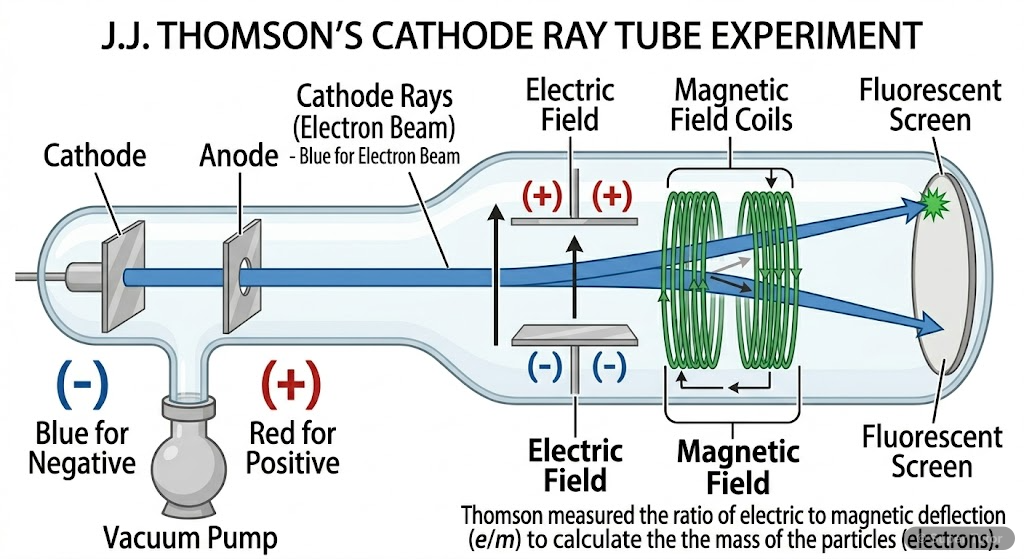
Learn better with visuals Super Tutor has hundreds of illustrations like this across every chapter — all free to try.
Get startedKey Topics to Revise
Discovery and Structure of Atom
- Ancient Indian concept: Maharshi Kanada (6th century BC) proposed 'paramanus' as ultimate particles
- Dalton's Atomic Theory (1808): Matter consists of indivisible atoms that combine in simple ratios
- J.J. Thomson discovered electrons through cathode ray experiments
Atomic Number, Mass Number and Electronic Configuration
- Atomic number (Z) = Number of protons = Number of electrons in neutral atom
- Mass number (A) = Number of protons + Number of neutrons
- Number of neutrons = Mass number - Atomic number
Isotopes and Chemical Activity
- Isotopes are atoms of same element with same atomic number but different mass numbers
- Isotopes have identical chemical properties but different physical properties
- Examples: Hydrogen (¹H, ²H, ³H), Carbon (¹²C, ¹³C, ¹⁴C), Chlorine (³⁵Cl, ³⁷Cl)
Electrovalent (Ionic) Bonding
- Formed by complete transfer of electrons from metal to non-metal
- Metal atoms lose electrons to form cations (positive ions)
- Non-metal atoms gain electrons to form anions (negative ions)
Get complete notes with diagrams and examples
Full NotesKey Concepts
Frequently Asked Questions
What are the important topics in Atomic Structure and Chemical Bonding for ICSE Class 9 Chemistry?
How to score full marks in Atomic Structure and Chemical Bonding — ICSE Class 9 Chemistry?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Atomic Structure and Chemical Bonding
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Study Plan
Step-by-step plan to ace this chapter
Flashcards
Quick-fire cards for active recall
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Atomic Structure and Chemical Bonding chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for ICSE Class 9 Chemistry.