The Periodic Table
ICSE · Class 9 · Chemistry
Quick revision notes for The Periodic Table — ICSE Class 9 Chemistry. Key concepts, formulas, and definitions for last-minute revision.
Interactive on Super Tutor
Studying The Periodic Table? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for revision notes and more.
1,000+ Class 9 students started this chapter today
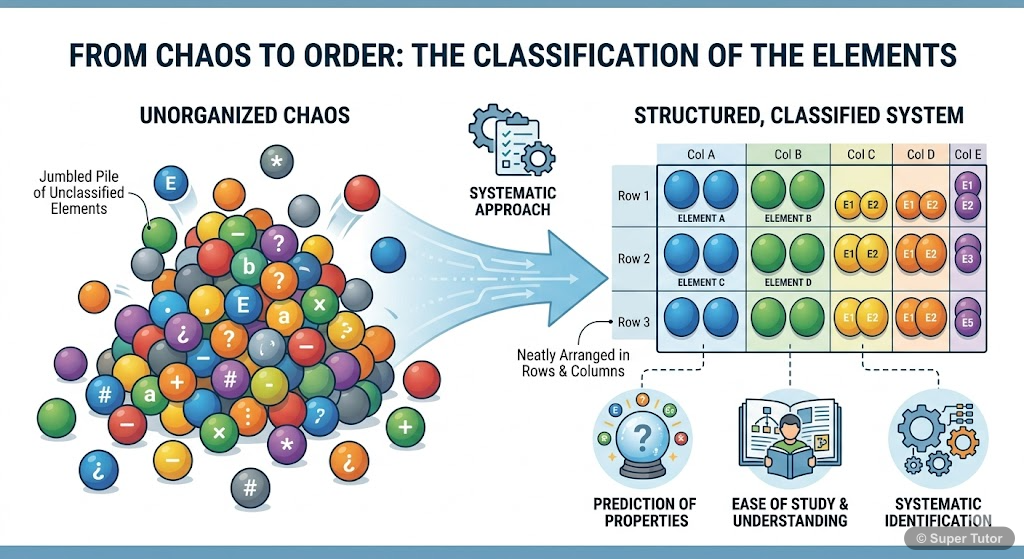
This is just one of 15+ visuals inside Super Tutor's The Periodic Table chapter
Explore the full setKey Topics to Revise
Historical Development of Periodic Classification
- Classification became necessary as more elements were discovered in the 18th and 19th centuries
- Early attempts based on density, malleability, and metallic/non-metallic nature were unsuccessful
- Dobereiner's Triads (1817): Elements grouped in sets of three with similar properties
Modern Periodic Law and Table Structure
- Henry Moseley (1913) discovered atomic number as the fundamental property
- Modern Periodic Law: Properties are periodic functions of atomic numbers
- Atomic number = number of protons in nucleus = number of electrons in neutral atom
Periodic Trends and Properties
- Down a group: atomic size increases, metallic character increases, reactivity of metals increases
- Across a period: atomic size decreases, metallic character decreases, valence electrons increase
- Valency with hydrogen: increases 1→4 then decreases 4→1 across period
Group 1: Alkali Metals
- Elements: Li, Na, K, Rb, Cs, Fr
- Electronic configuration: ns¹ (one valence electron)
- Highly reactive metals, reactivity increases down the group
Get complete notes with diagrams and examples
Full NotesKey Concepts
Frequently Asked Questions
What are the important topics in The Periodic Table for ICSE Class 9 Chemistry?
How to score full marks in The Periodic Table — ICSE Class 9 Chemistry?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for The Periodic Table
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Study Plan
Step-by-step plan to ace this chapter
Flashcards
Quick-fire cards for active recall
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full The Periodic Table chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for ICSE Class 9 Chemistry.