The d-and f-Block Elements
Karnataka Board · Class 12 · Chemistry
Summary of The d-and f-Block Elements for Karnataka Board Class 12 Chemistry. Key concepts, important points, and chapter overview.
Interactive on Super Tutor
Studying The d-and f-Block Elements? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for chapter summary and more.
1,000+ Class 12 students started this chapter today
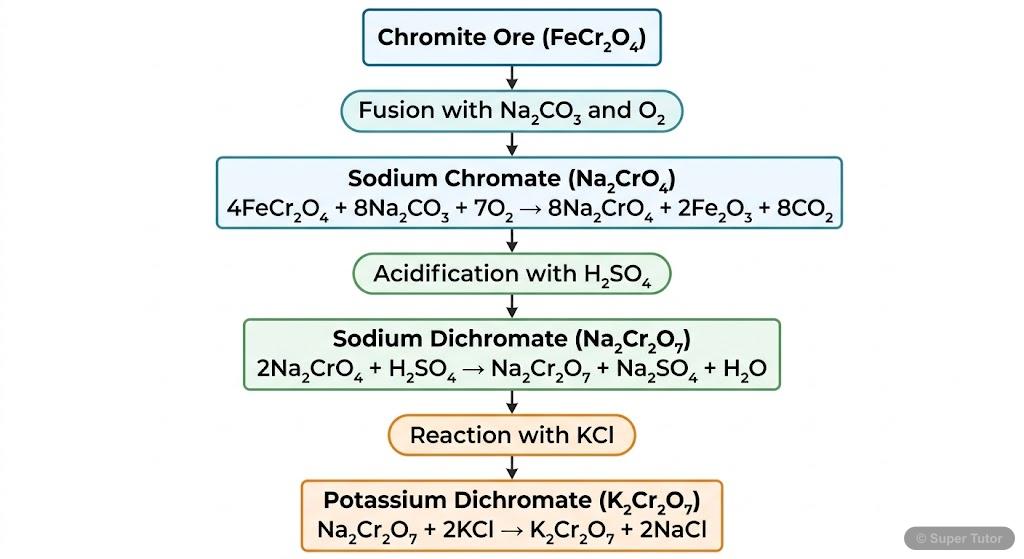
Super Tutor has 15+ illustrations like this for The d-and f-Block Elements alone — flashcards, concept maps, and step-by-step visuals.
See them allOverview
The d- and f-block elements, also known as transition and inner transition elements respectively, occupy the central portion of the periodic table and exhibit unique properties due to their partially filled d and f orbitals. These elements include some of the most important metals in human civilizat
Key Concepts
d
d-block elements (Groups 3-12) have general electronic configuration (n-1)d¹⁻¹⁰ns¹⁻² where d-orbitals are progressively filled. f-block elements have
Transition elements show multiple oxidation states
Transition elements show multiple oxidation states differing by unity (e.g., V²⁺, V³⁺, V⁴⁺, V⁵⁺) due to small energy differences between (n-1)d and ns
Progressive decrease in atomic and ionic
Progressive decrease in atomic and ionic radii across lanthanoid (4f filling) and actinoid (5f filling) series due to poor shielding by f-electrons. L
d¹
d¹-d⁹ configurations show colors due to d-d electronic transitions when electrons jump from lower to higher energy d-orbitals. Energy corresponds to v
Paramagnetic behavior due to unpaired d
Paramagnetic behavior due to unpaired d-electrons. Magnetic moment calculated using spin-only formula: μ = √n(n+2) BM where n = number of unpaired ele
Learning Objectives
- Understand the position and electronic configurations of d- and f-block elements in the periodic table
- Learn about the unique characteristics of transition elements including variable oxidation states, colored ion formation, and catalytic properties
- Analyze trends in atomic sizes, ionization enthalpies, and electrode potentials across transition series
- Study the preparation, properties, and applications of important compounds like K₂Cr₂O₇ and KMnO₄
- Compare and contrast lanthanoids and actinoids with respect to their electronic configurations, oxidation states, and chemical behavior
Frequently Asked Questions
What are the important topics in The d-and f-Block Elements for Karnataka Board Class 12 Chemistry?
How to score full marks in The d-and f-Block Elements — Karnataka Board Class 12 Chemistry?
Sources & Official References
- Karnataka SSLC — kseeb.kar.nic.in
- Dept of Pre-University Education, Karnataka
- National Education Policy 2020 — education.gov.in
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for The d-and f-Block Elements
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Flashcards
Quick-fire cards for active recall
Formula Sheet
All formulas in one place
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full The d-and f-Block Elements chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Karnataka Board Class 12 Chemistry.