Electrochemistry
Karnataka Board · Class 12 · Chemistry
Flashcards for Electrochemistry — Karnataka Board Class 12 Chemistry. Quick Q&A cards covering key concepts, definitions, and formulas.
Interactive on Super Tutor
Studying Electrochemistry? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for flashcards and more.
1,000+ Class 12 students started this chapter today
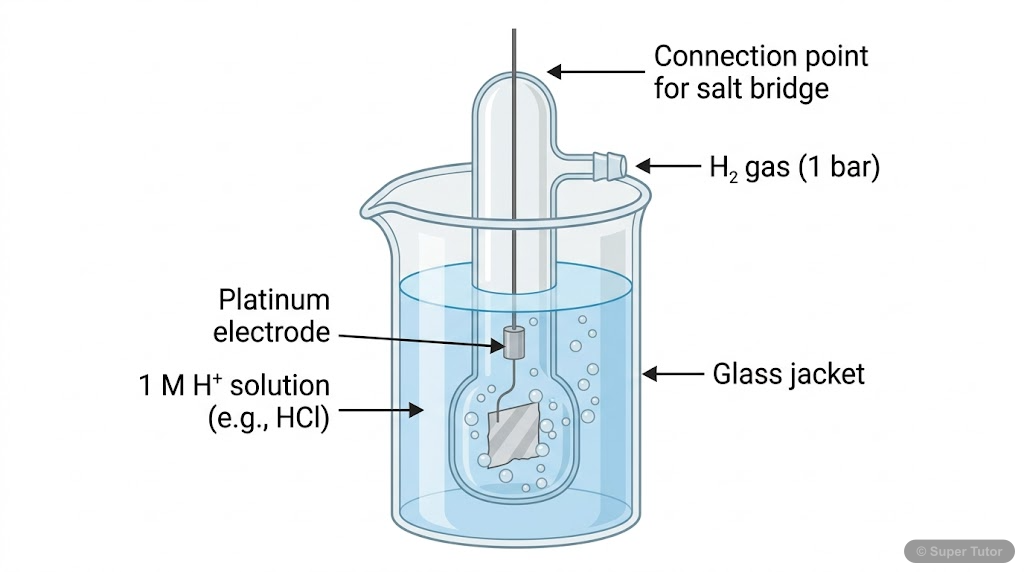
Learn better with visuals Super Tutor has hundreds of illustrations like this across every chapter — all free to try.
Get startedWhat is electrochemistry and what are its two main applications?
Answer
Electrochemistry is the study of the relationship between chemical reactions and electrical energy. Its two main applications are: (1) Production of electricity from spontaneous chemical reactions (ga…
Define galvanic cell and give an example with its cell reaction.
Answer
A galvanic cell converts chemical energy of a spontaneous redox reaction into electrical energy. Example: Daniell cell with Zn and Cu electrodes. Cell reaction: Zn(s) + Cu²⁺(aq) → Zn²⁺(aq) + Cu(s). Ce…
What is the difference between anode and cathode in a galvanic cell?
Answer
In a galvanic cell: Anode is the electrode where oxidation occurs (negative terminal, electrons are released). Cathode is the electrode where reduction occurs (positive terminal, electrons are consume…
What is standard electrode potential and how is it measured?
Answer
Standard electrode potential (E°) is the potential of an electrode when all species are at unit concentration (1 M) at 298 K and 1 bar pressure. It is measured relative to Standard Hydrogen Electrode …
Write the Nernst equation for a general electrode reaction Mⁿ⁺ + ne⁻ → M and explain its significance.
Answer
Nernst equation: E = E° - (RT/nF) ln(1/[Mⁿ⁺]) = E° - (0.059/n) log(1/[Mⁿ⁺]) at 298 K. Significance: It relates electrode potential to concentration of ions. Shows that potential decreases as concentra…
For the cell Zn|Zn²⁺||Cu²⁺|Cu, write the Nernst equation for cell potential.
Answer
E_cell = E°_cell - (RT/2F) ln([Zn²⁺]/[Cu²⁺]) = E°_cell - (0.059/2) log([Zn²⁺]/[Cu²⁺]) at 298 K. Where E°_cell = E°_Cu²⁺/Cu - E°_Zn²⁺/Zn = 0.34 - (-0.76) = 1.10 V.
How is Gibbs energy related to cell potential? Write the equation and its significance.
Answer
ΔrG = -nFE_cell. For standard conditions: ΔrG° = -nFE°_cell. Significance: Negative ΔrG means spontaneous reaction (positive E_cell). This relationship connects thermodynamics with electrochemistry an…
Define conductivity (κ) and molar conductivity (Λm). Give their units.
Answer
Conductivity (κ) = 1/ρ = conductance of 1 m³ solution between electrodes 1 m apart. Units: S m⁻¹ or S cm⁻¹. Molar conductivity (Λm) = κ/c = conductivity per unit concentration. Units: S m² mol⁻¹ or S …
+14 more flashcards available
Practice AllFrequently Asked Questions
What are the important topics in Electrochemistry for Karnataka Board Class 12 Chemistry?
How to score full marks in Electrochemistry — Karnataka Board Class 12 Chemistry?
How many flashcards are available for Electrochemistry?
Sources & Official References
- Karnataka SSLC — kseeb.kar.nic.in
- Dept of Pre-University Education, Karnataka
- National Education Policy 2020 — education.gov.in
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Electrochemistry
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Electrochemistry chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Karnataka Board Class 12 Chemistry.