Metals and Non-metals
Madhya Pradesh Board · Class 10 · Science
Flashcards for Metals and Non-metals — Madhya Pradesh Board Class 10 Science. Quick Q&A cards covering key concepts, definitions, and formulas.
Interactive on Super Tutor
Studying Metals and Non-metals? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for flashcards and more.
1,000+ Class 10 students started this chapter today
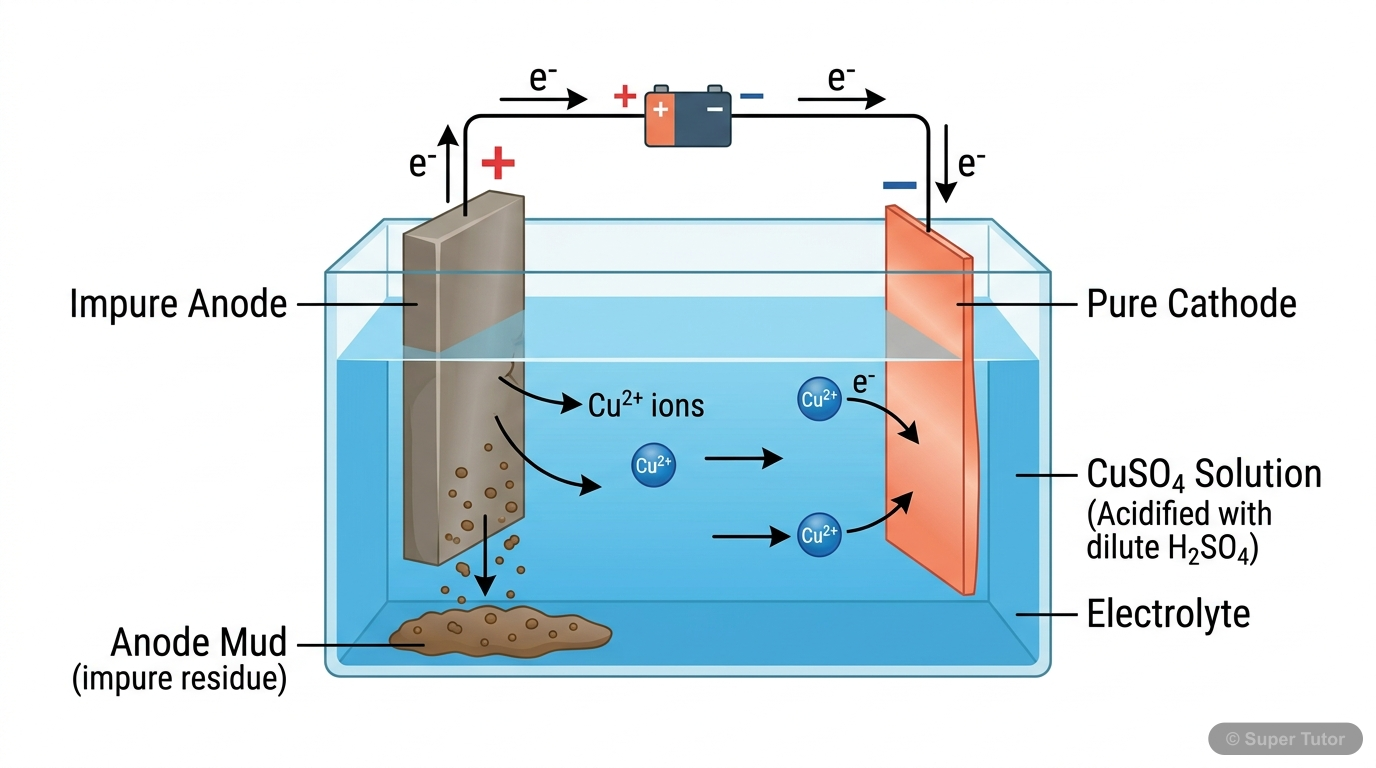
This is just one of 15+ visuals inside Super Tutor's Metals and Non-metals chapter
Explore the full setA student rubs a copper coin with sandpaper and observes it becomes shiny. What property of metals does this demonstrate?
Answer
This demonstrates metallic lustre - the property of metals to have a shining surface when freshly cut or cleaned. The sandpaper removes the oxide layer, revealing the shiny metallic surface underneath…
Why are cooking vessels made of aluminum or copper instead of wood or plastic?
Answer
Metals like aluminum and copper are good conductors of heat. They efficiently transfer heat from the flame to the food, ensuring even cooking. Wood and plastic are poor conductors and would burn or me…
What happens when sodium metal is dropped in water? Write the balanced chemical equation.
Answer
Sodium reacts violently with water, producing sodium hydroxide and hydrogen gas. The reaction is highly exothermic and the hydrogen catches fire. 2Na(s) + 2H₂O(l) → 2NaOH(aq) + H₂(g) + heat energy…
How can you test if a solution conducts electricity? What does this tell us about ionic compounds?
Answer
Set up a circuit with a bulb, battery, and two electrodes dipped in the solution. If the bulb glows, the solution conducts electricity. This shows ionic compounds dissociate into ions in solution, all…
Why do school bells produce sound when struck, but rubber balls do not?
Answer
School bells are made of metals which are sonorous - they produce sound when struck against hard surfaces. Rubber is a non-metal and lacks this property. The metallic structure allows vibrations to tr…
Explain why diamond is hard while graphite is soft, even though both are forms of carbon.
Answer
Both are allotropes of carbon with different structures. Diamond has a 3D network of carbon atoms with each carbon bonded to 4 others, making it extremely hard. Graphite has layered structure with wea…
How do you arrange these metals in decreasing order of reactivity: Iron, Copper, Magnesium, Zinc?
Answer
Based on the activity series: Magnesium > Zinc > Iron > Copper Magnesium reacts vigorously with acids and water, zinc and iron react with acids but not cold water, while copper doesn't react with dil…
A copper wire is placed in iron sulfate solution, and an iron nail in copper sulfate solution. In which test tube will you observe a reaction?
Answer
Reaction occurs in the test tube with iron nail in copper sulfate solution. Iron is more reactive than copper, so it displaces copper from copper sulfate: Fe(s) + CuSO₄(aq) → FeSO₄(aq) + Cu(s) The n…
+12 more flashcards available
Practice AllFrequently Asked Questions
What are the important topics in Metals and Non-metals for Madhya Pradesh Board Class 10 Science?
How to score full marks in Metals and Non-metals — Madhya Pradesh Board Class 10 Science?
How many flashcards are available for Metals and Non-metals?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Metals and Non-metals
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Metals and Non-metals chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Madhya Pradesh Board Class 10 Science.