Chemical Reaction and Equations
Madhya Pradesh Board · Class 10 · Science
Step-by-step guide to study Chemical Reaction and Equations in Madhya Pradesh Board Class 10 Science. Topics to cover, practice strategy, and time allocation.
Interactive on Super Tutor
Studying Chemical Reaction and Equations? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for study plan and more.
1,000+ Class 10 students started this chapter today
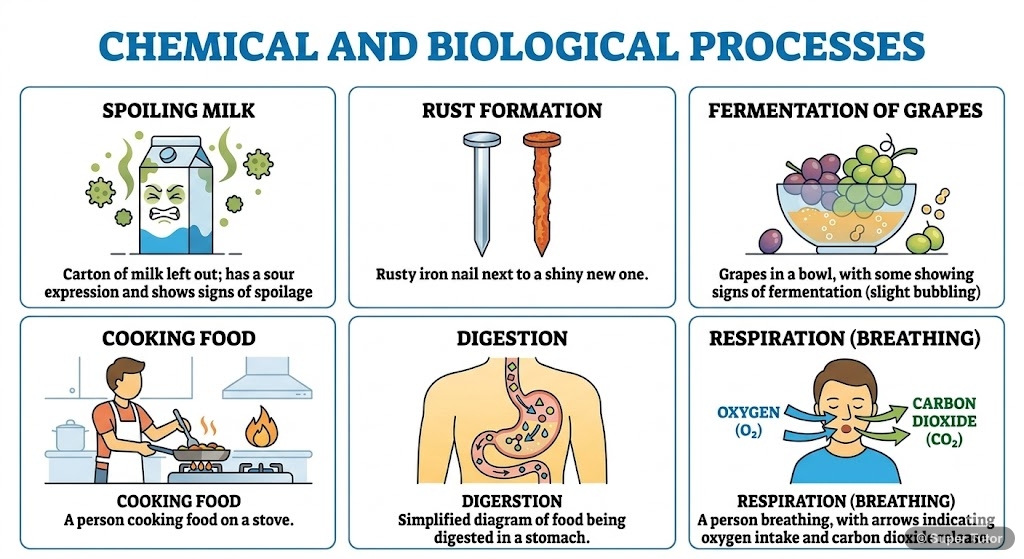
Super Tutor has 17+ illustrations like this for Chemical Reaction and Equations alone — flashcards, concept maps, and step-by-step visuals.
See them allStudy Plan
Learn the Theory
Read the textbook chapter carefully. Note down definitions, formulas, and key concepts.
Practice Problems
Solve textbook exercises and additional practice questions. There are 67 questions available for this chapter.
Revise & Test
Revise key formulas and concepts without looking at notes. Take a practice quiz to test your understanding. Mark weak areas for re-revision.
Spaced Revision
Revisit Chemical Reaction and Equations after a week. Use flashcards for quick recall. Solve previous year questions from this chapter.
What to Focus On
- Chemical reactions involve formation of new substances with different properties
- Atoms are rearranged but not created or destroyed
- Observable signs include color change, gas evolution, precipitate formation, and temperature change
- Chemical equations use symbols and formulas to represent reactions
- Reactants are written on the left, products on the right
- Arrow shows the direction of reaction
- Balancing follows the Law of Conservation of Mass
- Number of atoms of each element must be equal on both sides
- Use coefficients to balance, never change chemical formulas
Common Mistakes to Avoid
Balancing equations by changing chemical formulas instead of using coefficients
All combination reactions are exothermic and all decomposition reactions are endothermic
In displacement reactions, any metal can displace any other metal
Memory Tips
Signs of Chemical Reactions
Balancing Chemical Equations Steps
Types of Chemical Reactions
Combination Reaction
Want a personalised study plan?
Super Tutor creates a day-by-day plan for Madhya Pradesh Board Class 10 Science that adapts to your exam date and pace.
Create My Study Plan — FreeFrequently Asked Questions
What are the important topics in Chemical Reaction and Equations for Madhya Pradesh Board Class 10 Science?
How to score full marks in Chemical Reaction and Equations — Madhya Pradesh Board Class 10 Science?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Chemical Reaction and Equations
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Flashcards
Quick-fire cards for active recall
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Chemical Reaction and Equations chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Madhya Pradesh Board Class 10 Science.