Metals and Non-metals
Madhya Pradesh Board · Class 10 · Science
Complete topic list for Metals and Non-metals in Madhya Pradesh Board Class 10 Science. Key concepts, sub-topics, and what to focus on for board exams.
Interactive on Super Tutor
Studying Metals and Non-metals? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for syllabus and more.
1,000+ Class 10 students started this chapter today
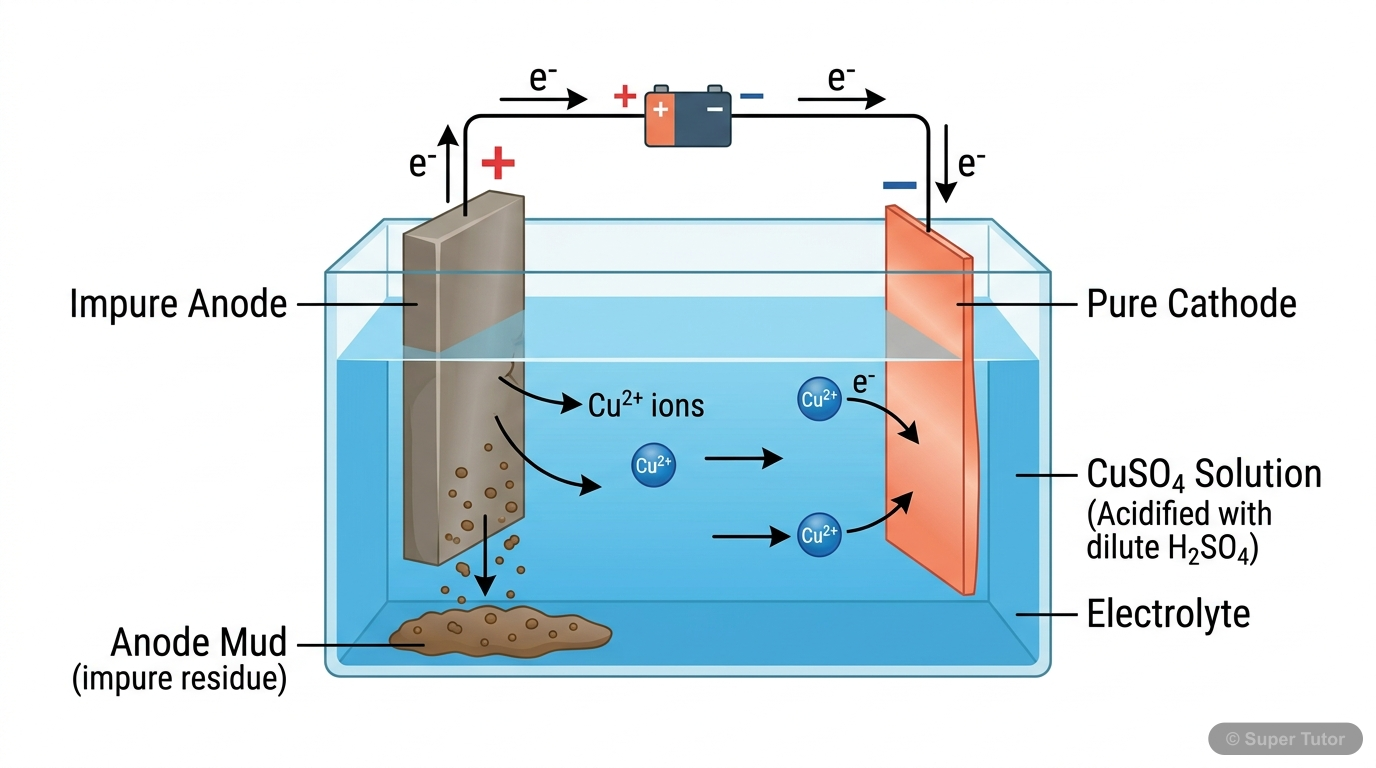
This is just one of 15+ visuals inside Super Tutor's Metals and Non-metals chapter
Explore the full setTopics in Metals and Non-metals
Physical Properties of Metals and Non-metals
- Metals have metallic lustre (shining surface) in pure state - iron, copper, aluminium show this property
- Malleability: Metals can be beaten into thin sheets (gold and silver are most malleable)
- Ductility: Metals can be drawn into thin wires (gold is most ductile - 2 km wire from 1g gold)
Chemical Properties and Reactivity of Metals
- Metals react with oxygen to form basic metal oxides (some like Al₂O₃, ZnO are amphoteric)
- Reactivity with oxygen: K, Na > Mg > Al > Zn > Fe > Pb > Cu > Hg > Ag, Au (no reaction)
- Metals react with water: K, Na (violent reaction with cold water), Ca (less violent), Mg (hot water only)
Ionic Compounds Formation and Properties
- Metals lose electrons to form positive ions (cations), non-metals gain electrons to form negative ions (anions)
- Ionic compounds form through electron transfer between metals and non-metals
- Na loses 1 electron to form Na⁺, Cl gains 1 electron to form Cl⁻, forming NaCl
Occurrence and Extraction of Metals
- Metals occur as minerals in earth's crust; profitable minerals are called ores
- Least reactive metals (Au, Ag, Pt, Cu) found in free state
- Moderately reactive metals (Zn, Fe, Pb) found as oxides, sulfides, carbonates
Key Concepts
Central concept: Metals and Non-metals
Frequently Asked Questions
What are the important topics in Metals and Non-metals for Madhya Pradesh Board Class 10 Science?
How to score full marks in Metals and Non-metals — Madhya Pradesh Board Class 10 Science?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Metals and Non-metals
Important Questions
Practice with board exam-style questions
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Flashcards
Quick-fire cards for active recall
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Metals and Non-metals chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Madhya Pradesh Board Class 10 Science.