Alcohols, Phenols and Ethers
Madhya Pradesh Board · Class 12 · Chemistry
Flashcards for Alcohols, Phenols and Ethers — Madhya Pradesh Board Class 12 Chemistry. Quick Q&A cards covering key concepts, definitions, and formulas.
Interactive on Super Tutor
Studying Alcohols, Phenols and Ethers? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for flashcards and more.
1,000+ Class 12 students started this chapter today
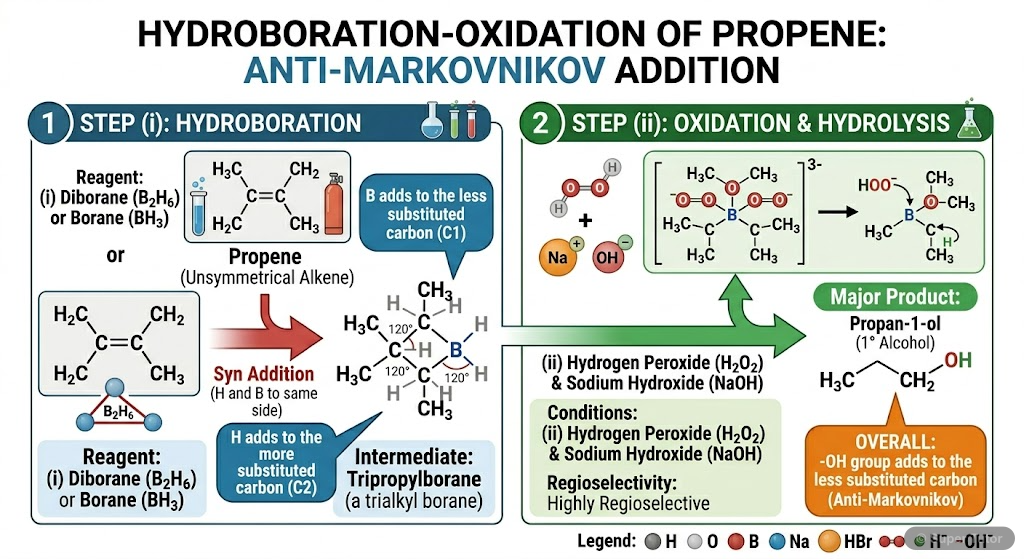
Super Tutor has 20+ illustrations like this for Alcohols, Phenols and Ethers alone — flashcards, concept maps, and step-by-step visuals.
See them allHow do you classify alcohols based on the carbon atom to which -OH is attached?
Answer
Primary (1°): -OH attached to carbon with one alkyl group (R-CH₂-OH) Secondary (2°): -OH attached to carbon with two alkyl groups (R₂CH-OH) Tertiary (3°): -OH attached to carbon with three alkyl group…
Write the balanced equation for acid-catalyzed hydration of propene to form an alcohol.
Answer
CH₃-CH=CH₂ + H₂O → CH₃-CH(OH)-CH₃ Reaction follows Markovnikov's rule: H⁺ adds to carbon with more hydrogens, OH⁻ adds to carbon with fewer hydrogens. Mechanism: Formation of 2° carbocation → nucleoph…
Calculate the number of moles of NaOH required to completely neutralize 0.1 mol of phenol (C₆H₅OH).
Answer
Balanced equation: C₆H₅OH + NaOH → C₆H₅ONa + H₂O Molar ratio: 1:1 (phenol : NaOH) Given: moles of phenol = 0.1 mol Moles of NaOH required = 0.1 mol Mass of NaOH = 0.1 × 40 = 4.0 g Answer: 0.1 moles of…
Why are phenols more acidic than alcohols? Explain with resonance structures.
Answer
Phenols are more acidic because the phenoxide ion (C₆H₅O⁻) is stabilized by resonance. Resonance structures show delocalization of negative charge over the benzene ring. In alcohols, alkoxide ion (RO⁻…
What products are formed when butan-2-ol reacts with concentrated H₂SO₄ at 170°C?
Answer
Dehydration reaction occurs: CH₃-CH(OH)-CH₂-CH₃ → CH₃-CH=CH-CH₃ + H₂O Major product: But-2-ene (follows Saytzeff's rule - most substituted alkene) Minor product: But-1-ene Mechanism: Protonation → car…
Calculate the volume of H₂ gas (at STP) produced when 2.3 g of ethanol reacts completely with sodium.
Answer
Balanced equation: 2C₂H₅OH + 2Na → 2C₂H₅ONa + H₂ Molar mass of ethanol = 46 g/mol Moles of ethanol = 2.3/46 = 0.05 mol From equation: 2 mol ethanol → 1 mol H₂ Moles of H₂ = 0.05/2 = 0.025 mol Volume a…
Write the complete mechanism for the Lucas test reaction with tertiary alcohols.
Answer
Lucas reagent: Conc. HCl + ZnCl₂ For tertiary alcohol: (CH₃)₃COH + HCl → (CH₃)₃CCl + H₂O Mechanism: 1. Protonation: (CH₃)₃COH + H⁺ → (CH₃)₃COH₂⁺ 2. Carbocation formation: (CH₃)₃COH₂⁺ → (CH₃)₃C⁺ + H₂O …
Name the following compounds according to IUPAC nomenclature: (i) CH₃CH(OH)CH₂CH₃ (ii) (CH₃)₂CHCH₂OH
Answer
(i) CH₃CH(OH)CH₂CH₃: Longest chain: 4 carbons (butane) -OH on carbon 2 IUPAC name: Butan-2-ol (ii) (CH₃)₂CHCH₂OH: Longest chain: 3 carbons (propane) Branched methyl on carbon 2 -OH on carbon 1 IUPAC …
+12 more flashcards available
Practice AllFrequently Asked Questions
What are the important topics in Alcohols, Phenols and Ethers for Madhya Pradesh Board Class 12 Chemistry?
How to score full marks in Alcohols, Phenols and Ethers — Madhya Pradesh Board Class 12 Chemistry?
How many flashcards are available for Alcohols, Phenols and Ethers?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Alcohols, Phenols and Ethers
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Alcohols, Phenols and Ethers chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Madhya Pradesh Board Class 12 Chemistry.