Chemical Kinetics
Madhya Pradesh Board · Class 12 · Chemistry
Practice quiz for Chemical Kinetics — Madhya Pradesh Board Class 12 Chemistry. MCQs and questions with answers to test your preparation.
Interactive on Super Tutor
Studying Chemical Kinetics? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for practice quiz and more.
1,000+ Class 12 students started this chapter today
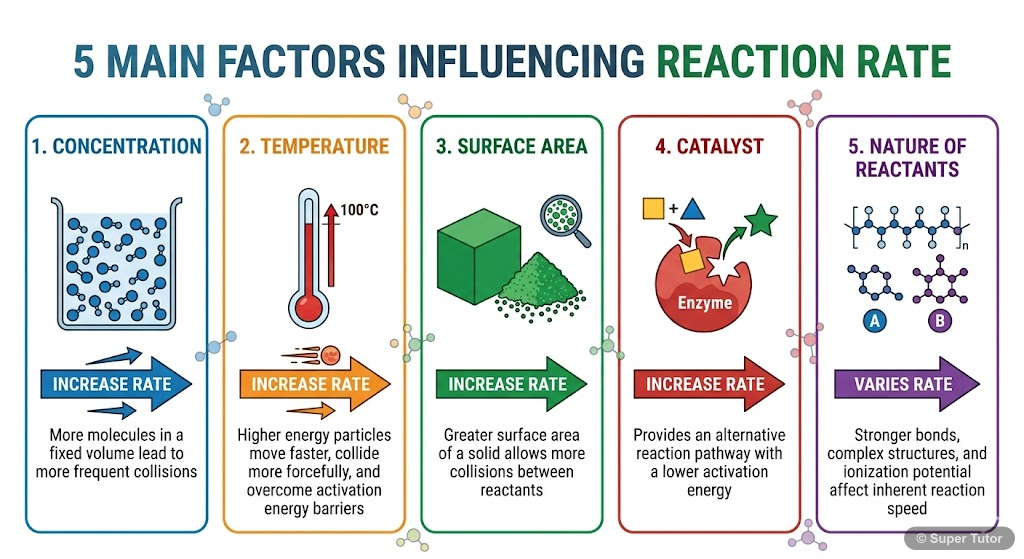
Learn better with visuals Super Tutor has hundreds of illustrations like this across every chapter — all free to try.
Get startedSample Questions
What is the unit of rate constant for a first-order reaction?
Which of the following factors affect the rate of a chemical reaction?
For a zero-order reaction, the half-life is:
The Arrhenius equation is k = Ae^(-Ea/RT). What does 'A' represent?
+28 more questions available
Practice AllFrequently Asked Questions
What are the important topics in Chemical Kinetics for Madhya Pradesh Board Class 12 Chemistry?
How to score full marks in Chemical Kinetics — Madhya Pradesh Board Class 12 Chemistry?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Chemical Kinetics
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Flashcards
Quick-fire cards for active recall
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Concept Maps
See how topics connect visually
For serious students
Get the full Chemical Kinetics chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Madhya Pradesh Board Class 12 Chemistry.