Carbon and its Compounds
Rajasthan Board · Class 10 · Science
Summary of Carbon and its Compounds for Rajasthan Board Class 10 Science. Key concepts, important points, and chapter overview.
Interactive on Super Tutor
Studying Carbon and its Compounds? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for chapter summary and more.
1,000+ Class 10 students started this chapter today
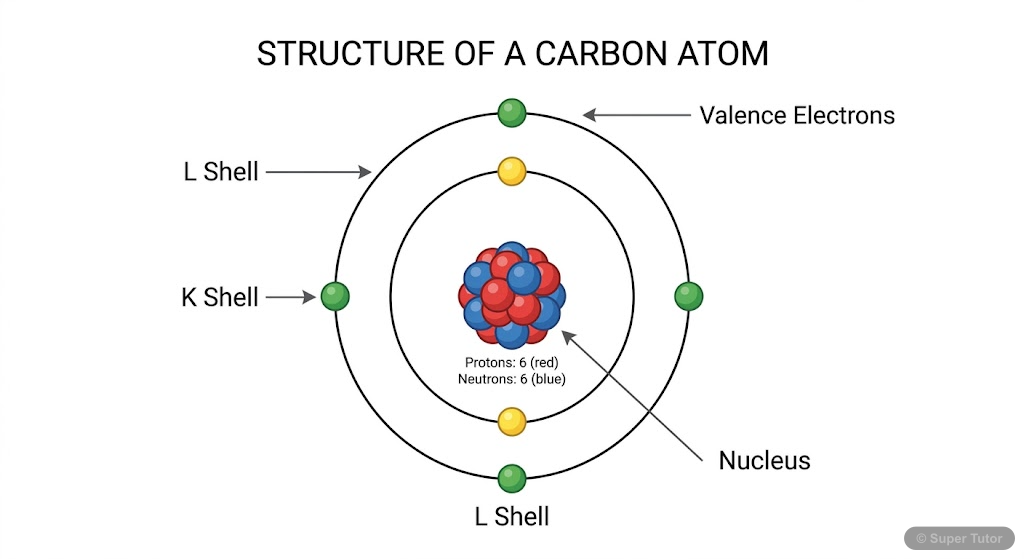
This is just one of 16+ visuals inside Super Tutor's Carbon and its Compounds chapter
Explore the full setOverview
Carbon is one of the most versatile elements in the periodic table, forming the backbone of all living organisms and countless useful compounds. Despite being only 0.02% of Earth's crust and 0.03% of the atmosphere, carbon's unique properties make it essential for life and technology. This chapter e
Key Concepts
Carbon has 4 valence electrons
Carbon has 4 valence electrons and needs 4 more to complete its octet. Instead of gaining or losing electrons (which would require too much energy), c
Carbon can form four covalent bonds
Carbon can form four covalent bonds as it has four valence electrons. This allows carbon to bond with four other atoms simultaneously, creating divers
Carbon's unique ability to form strong
Carbon's unique ability to form strong bonds with other carbon atoms, creating long chains, branched structures, and rings. Carbon-carbon bonds are ve
Saturated compounds contain only single bonds
Saturated compounds contain only single bonds between carbon atoms (alkanes like methane, ethane). Unsaturated compounds contain double or triple bond
Specific groups of atoms that replace
Specific groups of atoms that replace hydrogen in carbon compounds and give characteristic properties. Examples include alcohols (-OH), aldehydes (-CH
Learning Objectives
- Understand the concept of covalent bonding and how carbon forms covalent bonds
- Learn about carbon's versatile nature - tetravalency and catenation
- Explore different types of carbon compounds - saturated and unsaturated hydrocarbons
- Study the structure and naming of organic compounds and functional groups
- Understand chemical properties of carbon compounds including combustion, oxidation, addition, and substitution reactions
Frequently Asked Questions
What are the important topics in Carbon and its Compounds for Rajasthan Board Class 10 Science?
How to score full marks in Carbon and its Compounds — Rajasthan Board Class 10 Science?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Carbon and its Compounds
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Flashcards
Quick-fire cards for active recall
Formula Sheet
All formulas in one place
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Carbon and its Compounds chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Rajasthan Board Class 10 Science.