Carbon and its Compounds
Rajasthan Board · Class 10 · Science
Quick revision notes for Carbon and its Compounds — Rajasthan Board Class 10 Science. Key concepts, formulas, and definitions for last-minute revision.
Interactive on Super Tutor
Studying Carbon and its Compounds? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for revision notes and more.
1,000+ Class 10 students started this chapter today
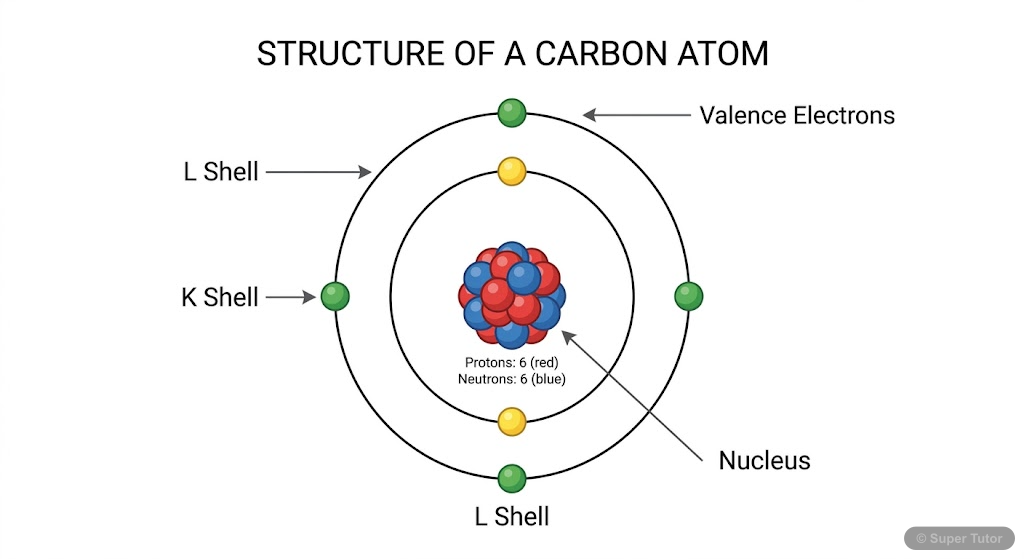
This is just one of 16+ visuals inside Super Tutor's Carbon and its Compounds chapter
Explore the full setKey Topics to Revise
Covalent Bonding and Carbon's Unique Properties
- Carbon has atomic number 6 with electronic configuration 2,4 - it has 4 valence electrons
- Carbon cannot form ionic bonds easily as gaining or losing 4 electrons requires too much energy
- Carbon shares electrons to form covalent bonds, achieving noble gas configuration
Types of Carbon Compounds and Structures
- Saturated compounds: Only single bonds between carbon atoms (alkanes)
- Unsaturated compounds: One or more double/triple bonds (alkenes, alkynes)
- Straight chain, branched chain, and cyclic structures are possible
Functional Groups and Nomenclature
- Major functional groups: -OH (alcohol), -CHO (aldehyde), -CO- (ketone), -COOH (carboxylic acid)
- Haloalkanes contain halogen atoms (Cl, Br, I) as functional groups
- Naming rules: Identify carbon chain length, then add prefix/suffix for functional group
Chemical Reactions of Carbon Compounds
- Combustion: Complete combustion produces CO₂ and H₂O with heat and light
- Incomplete combustion produces CO and soot (carbon particles)
- Saturated compounds burn with clean blue flame
Get complete notes with diagrams and examples
Full NotesKey Concepts
Frequently Asked Questions
What are the important topics in Carbon and its Compounds for Rajasthan Board Class 10 Science?
How to score full marks in Carbon and its Compounds — Rajasthan Board Class 10 Science?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Carbon and its Compounds
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Study Plan
Step-by-step plan to ace this chapter
Flashcards
Quick-fire cards for active recall
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Carbon and its Compounds chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Rajasthan Board Class 10 Science.