Solutions
Rajasthan Board · Class 12 · Chemistry
Flashcards for Solutions — Rajasthan Board Class 12 Chemistry. Quick Q&A cards covering key concepts, definitions, and formulas.
Interactive on Super Tutor
Studying Solutions? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for flashcards and more.
1,000+ Class 12 students started this chapter today
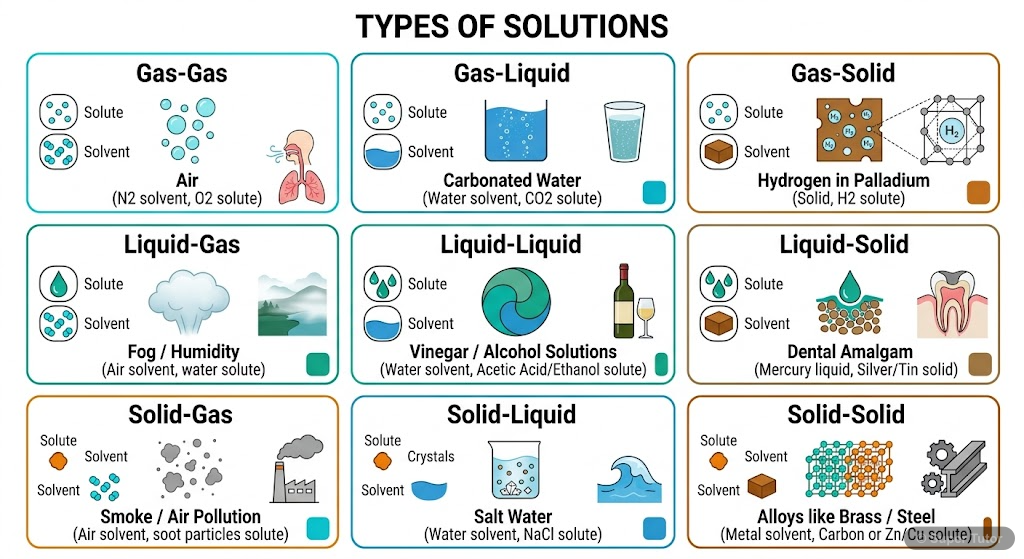
Super Tutor has 15+ illustrations like this for Solutions alone — flashcards, concept maps, and step-by-step visuals.
See them allDefine a solution and classify the different types based on physical states of solute and solvent.
Answer
A solution is a homogeneous mixture of two or more components with uniform composition and properties throughout. Types: Gaseous solutions (O₂ + N₂), Liquid solutions (glucose in water), Solid solutio…
Calculate the mass percentage of NaCl in a solution containing 20g NaCl dissolved in 180g water.
Answer
Formula: Mass % = (Mass of component/Total mass) × 100 Step 1: Total mass = 20g + 180g = 200g Step 2: Mass % of NaCl = (20/200) × 100 = 10% Answer: 10% by mass…
State Henry's Law and write its mathematical expression. Give one practical application.
Answer
Henry's Law: At constant temperature, the solubility of a gas in a liquid is directly proportional to the partial pressure of the gas above the solution. Mathematical form: p = KH × x Where p = partia…
Calculate molality of a solution containing 5.85g NaCl (M = 58.5 g/mol) dissolved in 500g water.
Answer
Formula: Molality (m) = moles of solute/kg of solvent Step 1: Moles of NaCl = 5.85/58.5 = 0.1 mol Step 2: Mass of water = 500g = 0.5 kg Step 3: Molality = 0.1/0.5 = 0.2 m Answer: 0.2 molal…
What is Raoult's Law? Write its equation for binary liquid solutions and explain ideal solutions.
Answer
Raoult's Law: The partial vapour pressure of each component in solution is directly proportional to its mole fraction. For binary solutions: p₁ = p₁⁰ × x₁ and p₂ = p₂⁰ × x₂ Total pressure: ptotal = p₁…
Calculate mole fraction of ethanol in a solution containing 46g ethanol (C₂H₅OH, M = 46 g/mol) and 54g water (M = 18 g/mol).
Answer
Step 1: Moles of ethanol = 46/46 = 1 mol Step 2: Moles of water = 54/18 = 3 mol Step 3: Total moles = 1 + 3 = 4 mol Step 4: Mole fraction of ethanol = 1/4 = 0.25 Answer: 0.25…
Why do non-ideal solutions show positive deviation from Raoult's Law? Give an example with explanation.
Answer
Positive deviation occurs when A-B interactions are weaker than A-A and B-B interactions. This makes molecules easier to escape, increasing vapour pressure above ideal value. Example: Ethanol + Aceton…
What are colligative properties? List all four with their definitions.
Answer
Colligative properties depend only on number of solute particles, not their nature. 1. Relative lowering of vapour pressure: (p₁⁰-p₁)/p₁⁰ = x₂ 2. Elevation of boiling point: ΔTb = Kb×m 3. Depression o…
+12 more flashcards available
Practice AllFrequently Asked Questions
What are the important topics in Solutions for Rajasthan Board Class 12 Chemistry?
How to score full marks in Solutions — Rajasthan Board Class 12 Chemistry?
How many flashcards are available for Solutions?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Solutions
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Solutions chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Rajasthan Board Class 12 Chemistry.