Structure of the Atom
Rajasthan Board · Class 9 · Science
Quick revision notes for Structure of the Atom — Rajasthan Board Class 9 Science. Key concepts, formulas, and definitions for last-minute revision.
Interactive on Super Tutor
Studying Structure of the Atom? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for revision notes and more.
1,000+ Class 9 students started this chapter today
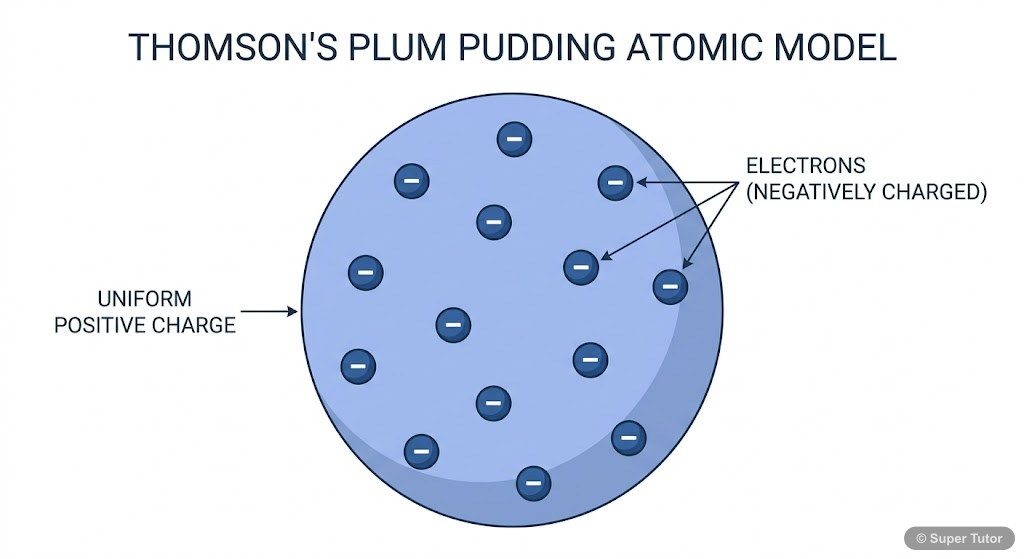
This is just one of 14+ visuals inside Super Tutor's Structure of the Atom chapter
Explore the full setKey Topics to Revise
Discovery of Sub-atomic Particles
- Atoms are not indivisible but contain smaller particles called sub-atomic particles
- Three main sub-atomic particles: electrons, protons, and neutrons
- J.J. Thomson discovered electrons in 1897 using cathode ray tube experiments
Atomic Models
- Thomson's Model (1904): 'Plum pudding' or 'watermelon' model - electrons embedded in positive sphere
- Rutherford's Model (1911): Nuclear model with electrons revolving around nucleus
- Rutherford's alpha particle scattering experiment revealed the nucleus
Electronic Configuration and Bohr-Bury Rules
- Electrons are arranged in shells or energy levels around the nucleus
- Shells are designated as K, L, M, N or n = 1, 2, 3, 4
- Maximum electrons in shell = 2n² where n is shell number
Valency
- Valency is the combining capacity of an atom
- Determined by the number of valence electrons
- Atoms tend to achieve stable electronic configuration (octet in outermost shell)
Get complete notes with diagrams and examples
Full NotesKey Concepts
Frequently Asked Questions
What are the important topics in Structure of the Atom for Rajasthan Board Class 9 Science?
How to score full marks in Structure of the Atom — Rajasthan Board Class 9 Science?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Structure of the Atom
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Study Plan
Step-by-step plan to ace this chapter
Flashcards
Quick-fire cards for active recall
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Structure of the Atom chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Rajasthan Board Class 9 Science.