Acids,Bases and Salts
Uttarakhand Board · Class 10 · Science
Summary of Acids,Bases and Salts for Uttarakhand Board Class 10 Science. Key concepts, important points, and chapter overview.
Interactive on Super Tutor
Studying Acids,Bases and Salts? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for chapter summary and more.
1,000+ Class 10 students started this chapter today
Super Tutor has 12+ illustrations like this for Acids,Bases and Salts alone — flashcards, concept maps, and step-by-step visuals.
See them allOverview
Acids and bases are fundamental chemical substances that we encounter in our daily lives. From the sourness of lemon juice to the bitter taste of soap, these substances have distinctive properties that make them easily identifiable. This chapter explores the chemical nature of acids and bases, their
Key Concepts
Acids are substances that produce hydrogen
Acids are substances that produce hydrogen ions (H+) when dissolved in water, making them sour in taste and turning blue litmus red. Bases produce hyd
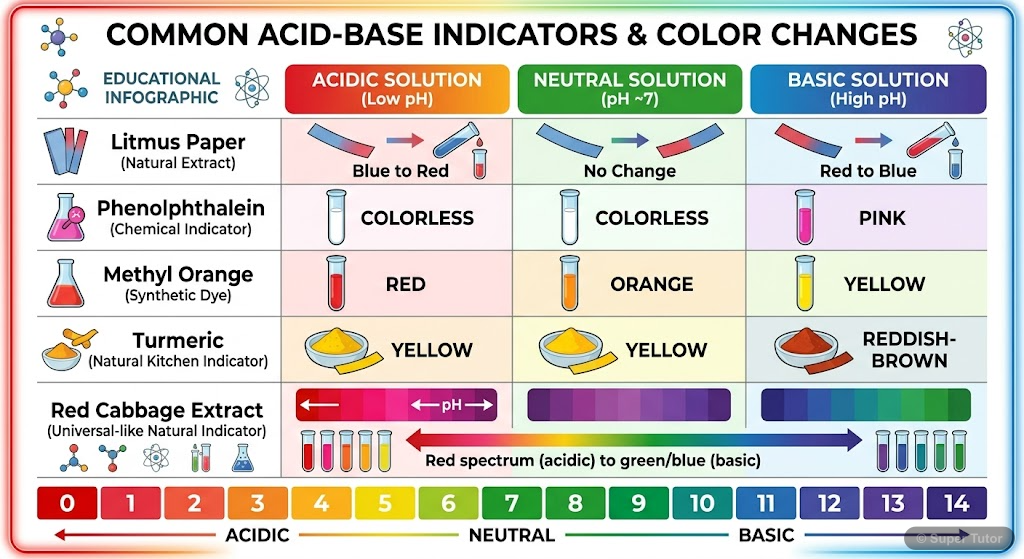
Indicators are substances that change color
Indicators are substances that change color in acidic or basic solutions. Natural indicators include litmus (from lichen), turmeric, and red cabbage.
Acids react with metals to produce
Acids react with metals to produce hydrogen gas and salts (Acid + Metal → Salt + H2). They react with metal carbonates and hydrogencarbonates to produ
The pH scale measures the concentration
The pH scale measures the concentration of hydrogen ions in a solution, ranging from 0 to 14. pH 7 is neutral, below 7 is acidic, and above 7 is basic
When an acid reacts with
When an acid reacts with a base, they neutralize each other to form salt and water (Acid + Base → Salt + Water). This reaction involves H+ ions from a
Learning Objectives
- Understand the chemical properties and behavior of acids and bases
- Learn how acids and bases react with metals, carbonates, and each other
- Explore the concept of pH and its importance in daily life
- Study the formation, types, and uses of salts
- Understand neutralization reactions and their applications
Frequently Asked Questions
What are the important topics in Acids,Bases and Salts for Uttarakhand Board Class 10 Science?
How to score full marks in Acids,Bases and Salts — Uttarakhand Board Class 10 Science?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Acids,Bases and Salts
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Flashcards
Quick-fire cards for active recall
Formula Sheet
All formulas in one place
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Acids,Bases and Salts chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Uttarakhand Board Class 10 Science.