Coordination Compounds
Uttarakhand Board · Class 12 · Chemistry
Flashcards for Coordination Compounds — Uttarakhand Board Class 12 Chemistry. Quick Q&A cards covering key concepts, definitions, and formulas.
Interactive on Super Tutor
Studying Coordination Compounds? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for flashcards and more.
1,000+ Class 12 students started this chapter today
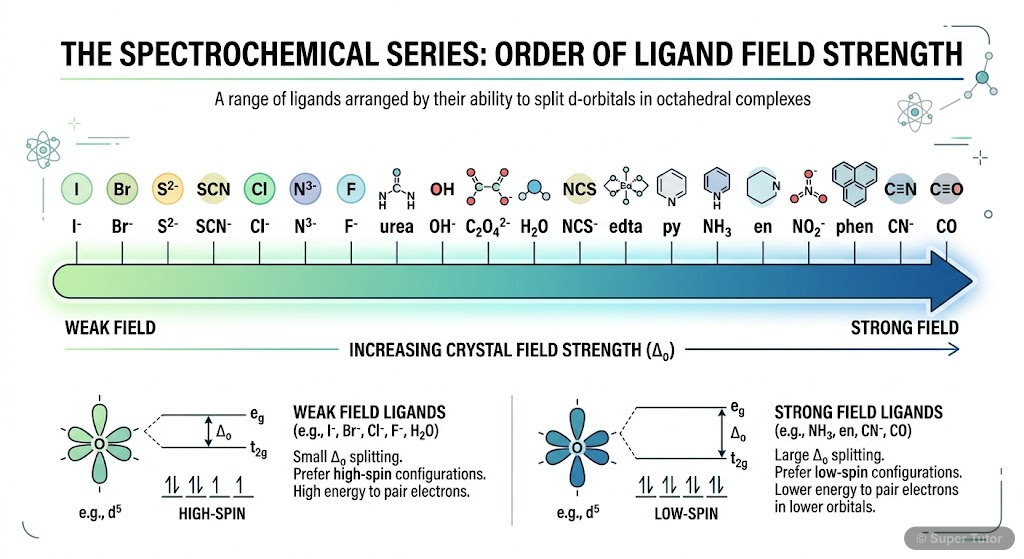
Super Tutor has 13+ illustrations like this for Coordination Compounds alone — flashcards, concept maps, and step-by-step visuals.
See them allAccording to Werner's theory, what are primary and secondary valences? Give an example with CoCl₃·6NH₃.
Answer
Primary valence = ionizable bonds (oxidation state). Secondary valence = non-ionizable bonds (coordination number). Example: [Co(NH₃)₆]Cl₃ - Co³⁺ has primary valence 3 (satisfied by 3Cl⁻) and secondar…
Calculate the oxidation state of chromium in [Cr(NH₃)₄Cl₂]Cl
Answer
Step 1: Identify charges - NH₃ = 0, Cl⁻ = -1, overall charge = +1 Step 2: Let oxidation state of Cr = x Step 3: x + 4(0) + 2(-1) = +1 Step 4: x - 2 = +1 Step 5: x = +3 Answer: Chromium is in +3 oxidat…
What is the difference between coordination number and oxidation number? Explain with [Fe(CN)₆]³⁻.
Answer
Coordination number = number of ligand donor atoms directly bonded to metal. Oxidation number = charge on metal if all ligands removed. In [Fe(CN)₆]³⁻: Coordination number = 6 (six CN⁻ ligands), Oxida…
Write the formation reaction of [Cu(NH₃)₄]²⁺ complex and identify the Lewis acid and Lewis base.
Answer
Cu²⁺(aq) + 4NH₃(aq) → [Cu(NH₃)₄]²⁺(aq) Lewis acid: Cu²⁺ (electron pair acceptor) Lewis base: NH₃ (electron pair donor) This is a coordination complex formation where NH₃ donates lone pairs to vacant o…
Name the compound [Co(en)₂Cl₂]Cl using IUPAC nomenclature rules.
Answer
dichlorido-bis(ethane-1,2-diamine)cobalt(III) chloride Rules applied: 1) Ligands in alphabetical order (chlorido before ethane-1,2-diamine), 2) bis- for complex ligand names, 3) Roman numeral for oxid…
What are chelating ligands? Why do they form more stable complexes? Give two examples.
Answer
Chelating ligands = polydentate ligands that bind through 2 or more donor atoms to same metal ion. More stable due to chelate effect (entropy increase when multiple monodentate ligands replaced). Exam…
Calculate the number of unpaired electrons in [CoF₆]³⁻ and predict if it's diamagnetic or paramagnetic.
Answer
Step 1: Co³⁺ configuration = 3d⁶ Step 2: F⁻ is weak field ligand → high spin Step 3: Electron arrangement in octahedral field: t₂g: ↑↓ ↑ ↑ eg: ↑ ↑ Step 4: Unpaired electrons = 4 Step 5: Paramagnetic (…
Why is [Co(NH₃)₆]³⁺ diamagnetic while [CoF₆]³⁻ is paramagnetic? Explain using crystal field theory.
Answer
Both contain Co³⁺ (3d⁶). NH₃ is strong field ligand causing large Δₒ > P (pairing energy), forcing electrons to pair: t₂g⁶eg⁰ → diamagnetic. F⁻ is weak field ligand with small Δₒ < P, electrons remain…
+12 more flashcards available
Practice AllFrequently Asked Questions
What are the important topics in Coordination Compounds for Uttarakhand Board Class 12 Chemistry?
How to score full marks in Coordination Compounds — Uttarakhand Board Class 12 Chemistry?
How many flashcards are available for Coordination Compounds?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Coordination Compounds
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Coordination Compounds chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Uttarakhand Board Class 12 Chemistry.