Chemical Reaction and Equations
Uttar Pradesh Board · Class 10 · Science
Summary of Chemical Reaction and Equations for Uttar Pradesh Board Class 10 Science. Key concepts, important points, and chapter overview.
Interactive on Super Tutor
Studying Chemical Reaction and Equations? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for chapter summary and more.
1,000+ Class 10 students started this chapter today
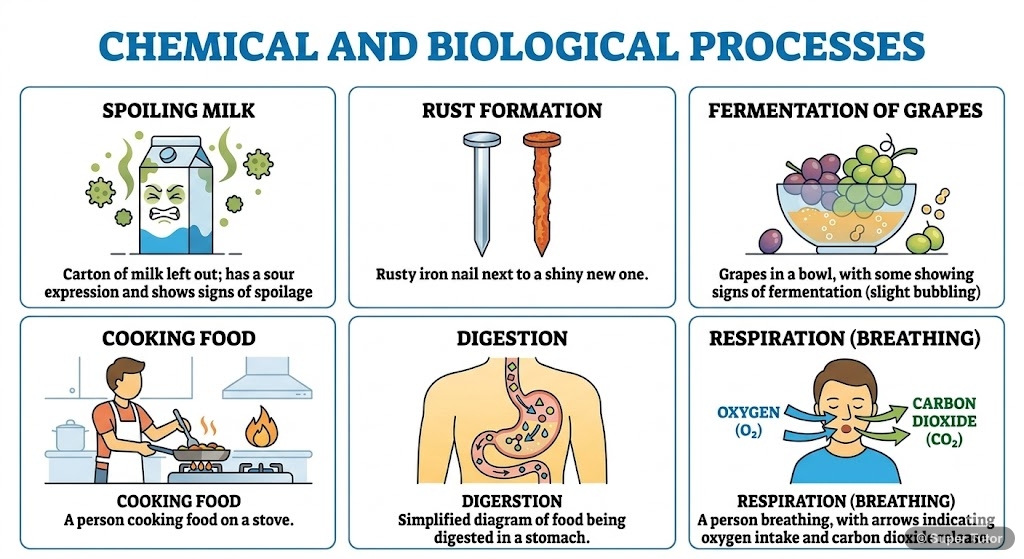
Super Tutor has 17+ illustrations like this for Chemical Reaction and Equations alone — flashcards, concept maps, and step-by-step visuals.
See them allOverview
Chemical reactions are fundamental processes that occur all around us in daily life. When milk sours, iron rusts, food cooks, or we breathe, chemical reactions are taking place. A chemical reaction involves the transformation of one or more substances (reactants) into new substances (products) with
Key Concepts
A process in which one
A process in which one or more substances undergo chemical change to form new substances with different properties. Signs include change in color, sta
A symbolic representation of a chemical
A symbolic representation of a chemical reaction using chemical formulae. Shows reactants on the left, products on the right, separated by an arrow. M
An equation where the number
An equation where the number of atoms of each element is equal on both reactant and product sides, following the law of conservation of mass.
A reaction where two or more
A reaction where two or more substances combine to form a single product. Example: 2H₂ + O₂ → 2H₂O. Often exothermic.
A reaction where a single reactant
A reaction where a single reactant breaks down into two or more simpler products. Can be thermal, electrolytic, or photolytic. Often endothermic.
Learning Objectives
- Understand what constitutes a chemical reaction and identify signs of chemical change
- Learn to write and balance chemical equations using proper symbols and formulae
- Classify different types of chemical reactions: combination, decomposition, displacement, and double displacement
- Distinguish between exothermic and endothermic reactions
- Understand oxidation and reduction processes in chemical reactions
Frequently Asked Questions
What are the important topics in Chemical Reaction and Equations for Uttar Pradesh Board Class 10 Science?
How to score full marks in Chemical Reaction and Equations — Uttar Pradesh Board Class 10 Science?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Chemical Reaction and Equations
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Flashcards
Quick-fire cards for active recall
Formula Sheet
All formulas in one place
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Chemical Reaction and Equations chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Uttar Pradesh Board Class 10 Science.