Structure of the Atom
Uttar Pradesh Board · Class 9 · Science
Flashcards for Structure of the Atom — Uttar Pradesh Board Class 9 Science. Quick Q&A cards covering key concepts, definitions, and formulas.
Interactive on Super Tutor
Studying Structure of the Atom? Get the full interactive chapter.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan — built for flashcards and more.
1,000+ Class 9 students started this chapter today
This is just one of 14+ visuals inside Super Tutor's Structure of the Atom chapter
Explore the full setWhat are the three fundamental sub-atomic particles found in an atom?
Answer
The three fundamental sub-atomic particles are: 1. Electrons (e⁻) - negatively charged, negligible mass 2. Protons (p⁺) - positively charged, mass = 1 unit 3. Neutrons (n) - no charge, mass = 1 unit…
Who discovered the electron and in which year?
Answer
J.J. Thomson discovered the electron. He was awarded the Nobel Prize in Physics in 1906 for his work on the discovery of electrons.
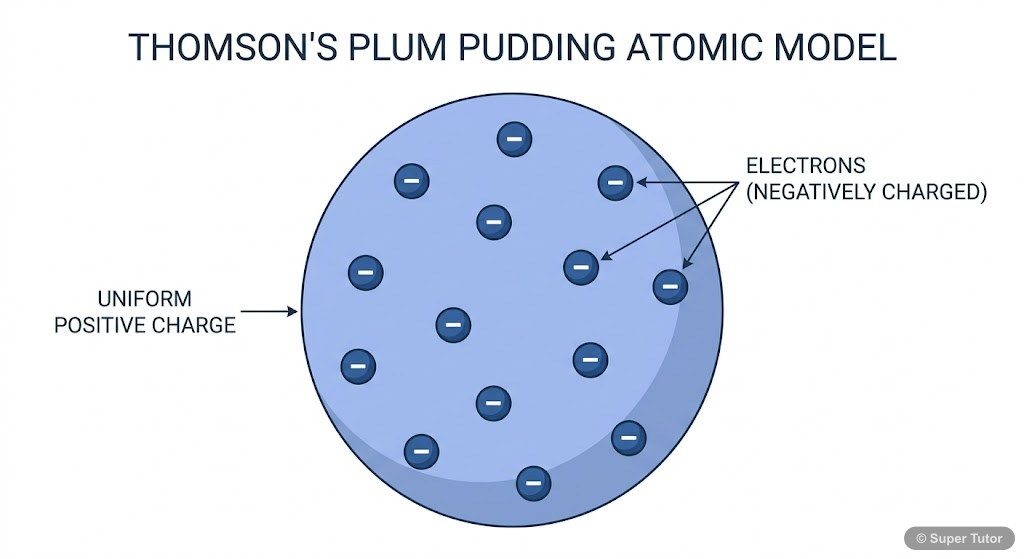
Describe Thomson's model of the atom.
Answer
Thomson's model (Christmas pudding model): - Atom consists of a positively charged sphere - Electrons are embedded in this positive sphere like currants in a pudding - Negative and positive charges ar…
What were the key observations from Rutherford's gold foil experiment?
Answer
Key observations: 1. Most α-particles passed straight through the gold foil 2. Some α-particles were deflected by small angles 3. Very few (1 in 12,000) α-particles rebounded (deflected by 180°) This…
What conclusions did Rutherford draw from his experiment?
Answer
Rutherford's conclusions: 1. Most of the atom is empty space 2. Positive charge occupies very little space 3. All positive charge and mass are concentrated in a tiny nucleus 4. Nucleus radius is 10⁵ t…
What was the major drawback of Rutherford's atomic model?
Answer
The major drawback was that revolving electrons should radiate energy continuously and spiral into the nucleus, making atoms unstable. However, atoms are actually stable, so this model could not expla…
State the main postulates of Bohr's atomic model.
Answer
Bohr's postulates: 1. Only certain special orbits (discrete orbits) are allowed for electrons 2. Electrons do not radiate energy while revolving in these discrete orbits 3. These orbits are called ene…
Who discovered neutrons and in which year?
Answer
J. Chadwick discovered neutrons in 1932. Neutrons have no charge and mass nearly equal to protons. They are present in the nucleus of all atoms except hydrogen.
+16 more flashcards available
Practice AllFrequently Asked Questions
What are the important topics in Structure of the Atom for Uttar Pradesh Board Class 9 Science?
How to score full marks in Structure of the Atom — Uttar Pradesh Board Class 9 Science?
How many flashcards are available for Structure of the Atom?
Sources & Official References
Content is aligned to the official syllabus. Refer to the board website for the latest curriculum.
More resources for Structure of the Atom
Important Questions
Practice with board exam-style questions
Syllabus
What topics to cover
Revision Notes
Key points for last-minute revision
Study Plan
Step-by-step plan to ace this chapter
Formula Sheet
All formulas in one place
Chapter Summary
Understand the chapter at a glance
Practice Quiz
Test yourself with a quick quiz
Concept Maps
See how topics connect visually
For serious students
Get the full Structure of the Atom chapter — for free.
Quizzes, flashcards, AI doubt-solver and a step-by-step study plan for Uttar Pradesh Board Class 9 Science.