Organic Chemistry Some Basic Principles and Techniques — Revision Notes
CG PET · Chemistry
Quick revision notes for Organic Chemistry Some Basic Principles and Techniques — key concepts, formulas, and definitions for CG PET Chemistry preparation.
Interactive on Super Tutor
Studying Organic Chemistry Some Basic Principles and Techniques? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for CG PET Chemistry.
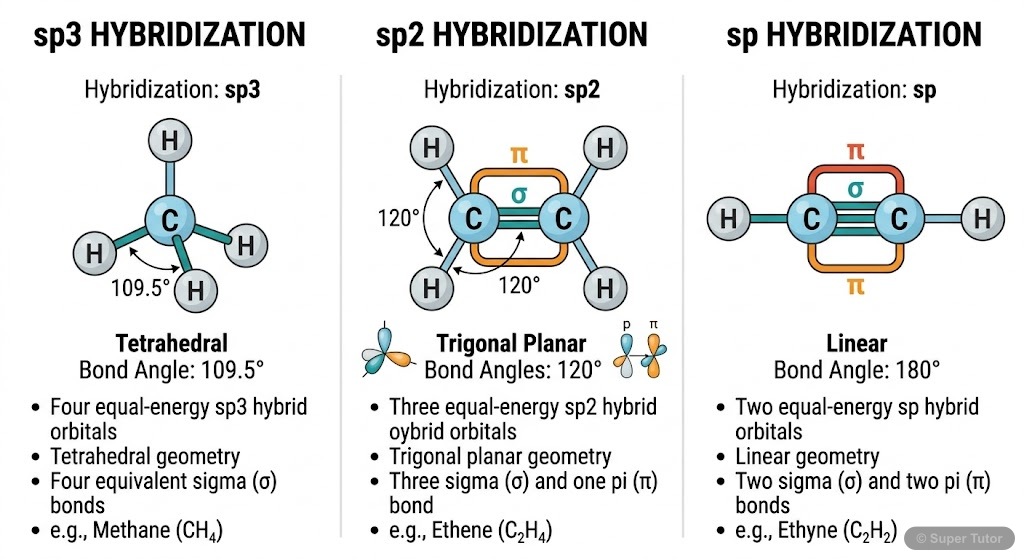
This is just one of 23+ visuals inside Super Tutor's Organic Chemistry Some Basic Principles and Techniques chapter
Explore the full setRevision Notes — Organic Chemistry Some Basic Principles and Techniques
Key concepts, formulas, and definitions from Organic Chemistry Some Basic Principles and Techniques for CG PET Chemistry preparation.
Key Topics to Revise
General Introduction and Tetravalence of Carbon
- Organic chemistry studies hydrocarbons and their derivatives containing C, H, N, O, S, P, halogens
- Berzelius proposed 'vital force' theory - later disproved by Wöhler's urea synthesis
- NH₄CNO → NH₂-CO-NH₂ (Wöhler, 1828) - first organic compound from inorganic source
Structural Representations and Classification
- Complete structural formulas show all atoms and bonds explicitly
- Condensed formulas omit bonds but show groupings with subscripts
- Bond-line formulas represent carbon skeleton as lines with functional groups
IUPAC Nomenclature
- IUPAC name = Secondary prefix + Primary prefix + Word root + Primary suffix + Secondary suffix
- Functional group priority: -COOH > -SO₃H > -COOR > -COCl > -CONH₂ > -CN > -CHO > >C=O > -OH > -NH₂
- Longest carbon chain containing principal functional group is parent chain
Isomerism
- Isomerism: same molecular formula but different properties
- Structural isomerism: different connectivity - chain, position, functional, metamerism, tautomerism
- Stereoisomerism: same connectivity but different spatial arrangement
Get complete revision notes with diagrams and examples — continue in Super Tutor
Key Concepts
Frequently Asked Questions
What topics are covered in Organic Chemistry Some Basic Principles and Techniques for CG PET?
How important is Organic Chemistry Some Basic Principles and Techniques for CG PET?
How to prepare Organic Chemistry Some Basic Principles and Techniques for CG PET?
More resources for Organic Chemistry Some Basic Principles and Techniques
Syllabus
Organic Chemistry Some Basic Principles and Techniques — syllabus
Important Topics
Organic Chemistry Some Basic Principles and Techniques — important topics
Practice Questions
Organic Chemistry Some Basic Principles and Techniques — practice questions
Study Plan
Organic Chemistry Some Basic Principles and Techniques — study plan
Formula Sheet
Organic Chemistry Some Basic Principles and Techniques — formula sheet
For CG PET aspirants
Get the full Organic Chemistry Some Basic Principles and Techniques chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for CG PET Chemistry.