Chapter 11 of 22
Revision Notes
Solutions — Revision Notes
NEET-UG · Chemistry
Quick revision notes for Solutions — key concepts, formulas, and definitions for NEET-UG Chemistry preparation.
Interactive on Super Tutor
Studying Solutions? Get the full chapter — free.
Practice questions, revision notes, formula sheet and AI doubt-solver — built for NEET-UG Chemistry.
Super Tutor
This is just one of 17+ visuals inside Super Tutor's Solutions chapter
Explore the full setRevision Notes — Solutions
Key concepts, formulas, and definitions from Solutions for NEET-UG Chemistry preparation.
Key Topics to Revise
1
Types of Solutions and Concentration Units
- Solutions can be gaseous (O₂ + N₂), liquid (ethanol in water), or solid (brass alloy)
- Mass percentage = (mass of solute/mass of solution) × 100
- Volume percentage used for liquid-liquid solutions like alcohol-water mixtures
2
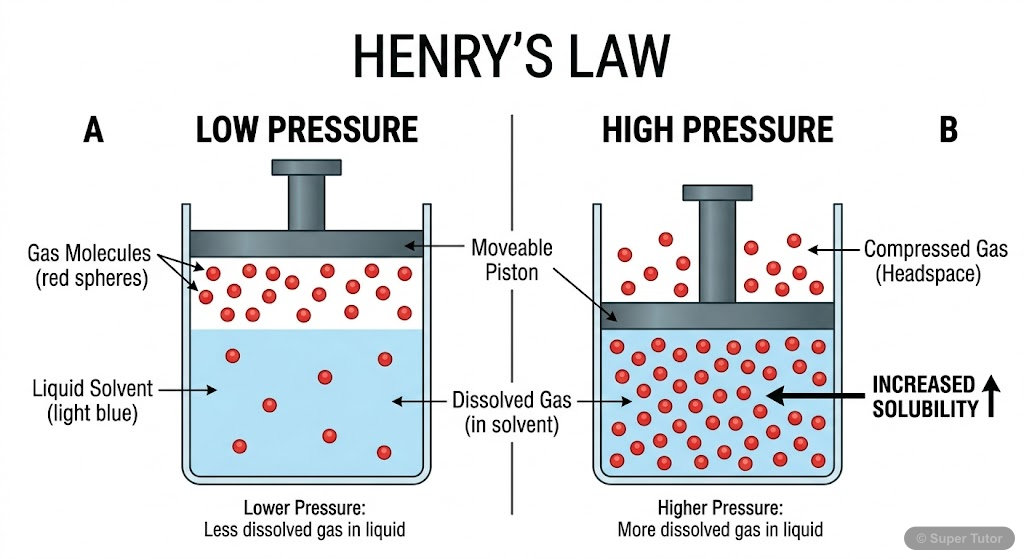
Henry's Law and Gas Solubility
- Henry's law: Solubility of gas ∝ pressure of gas above solution
- Mathematical form: m = KP or p = KHχ where KH is Henry's constant
- Higher KH value means lower gas solubility in liquid
3
Raoult's Law and Vapor Pressure
- Raoult's law: PA = χA × P°A for each component in solution
- Total pressure: PT = PA + PB = χA·P°A + χB·P°B
- Vapor pressure lowering: ΔP = χsolute × P°solvent
4
Colligative Properties - Boiling Point Elevation and Freezing Point Depression
- Colligative properties depend only on number of solute particles, not their identity
- Boiling point elevation: ΔTb = Kb × m where Kb is ebullioscopic constant
- Freezing point depression: ΔTf = Kf × m where Kf is cryoscopic constant
Get complete revision notes with diagrams and examples — continue in Super Tutor
Key Concepts
Solutions can be classified based onMass % = (mass of solute/massThe solubility of a gasFor ideal solutionsIdeal solutions obey Raoult's law
Frequently Asked Questions
What topics are covered in Solutions for NEET-UG?
Solutions is an important chapter in NEET-UG Chemistry. It covers key concepts and formulas that are frequently tested in the exam. Key topics include: Types of Solutions and Concentration Units, Henry's Law and Gas Solubility, Raoult's Law and Vapor Pressure, Colligative Properties - Boiling Point Elevation and Freezing Point Depression.
How important is Solutions for NEET-UG?
Solutions is a frequently tested chapter in NEET-UG Chemistry. Questions from this chapter appear regularly in previous year papers. There are 81 practice questions available for this chapter.
How to prepare Solutions for NEET-UG?
Start by understanding the core concepts, then solve practice questions. Focus on formulas and their applications. Use revision notes for quick review before the exam.
More resources for Solutions
For NEET-UG aspirants
Get the full Solutions chapter — for free.
Practice questions, revision notes, formula sheet and AI doubt-solver for NEET-UG Chemistry.